Research Article | DOI: https://doi.org/10.31579/2835-8295/160
The Content of Heat Shock Protein Hsp 70 In Neurons of the Parietal Cortex and Hippocampus of Rats with Cerebral Ischemia of Varying Severity
- E.I. Bon , *
- N.Ye. Maksimovich,
- O.A. Karnyushko,
- S.M. Zimatkin, A.I,
- Otlivanchick
Candidate of Biological Sciences, Associate Professor of the Department of Pathological Physiology named D.A. Maslakova Grodno State Medical University, Grodno, Republic of Belarus.
*Corresponding Author: E.I. Bon, Candidate of Biological Sciences, Associate Professor of the Department of Pathological Physiology named D.A. Maslakova Grodno State Medical University, Grodno, Republic of Belarus.
Citation: E.I. Bon, N.Ye. Maksimovich, O.A. Karnyushko, S.M. Zimatkin, A.I. Otlivanchick, (2026), The Content of Heat Shock Protein Hsp 70 In Neurons of the Parietal Cortex and Hippocampus of Rats with Cerebral Ischemia of Varying Severity, International Journal of Clinical Reports and Studies, 5(3); DOI:10.31579/2835-8295/160
Copyright: © 2026, E.I. Bon. This is an open-access artic le distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 27 April 2026 | Accepted: 04 May 2026 | Published: 15 May 2026
Keywords: cerebrovascular; cardiovascular; hypoxia
Abstract
Heat shock proteins (English HSP, Heat shock proteins) are a class of functionally similar proteins, the expression of which increases with increasing temperature or other stress effects on the cell, including ischemia [8]. Heat shock proteins are universal molecular chaperones (from English chaperon – to accompany), i.e. proteins that bind to other molecules and perform certain functions in such a complex
Introduction
Heat shock proteins (English HSP, Heat shock proteins) are a class of functionally similar proteins, the expression of which increases with increasing temperature or other stress effects on the cell, including ischemia [8]. Heat shock proteins are universal molecular chaperones (from English chaperon – to accompany), i.e. proteins that bind to other molecules and perform certain functions in such a complex [14]. The main function of HSP is considered to be the control of the formation of new proteins and the formation of their tertiary structure (folding). By binding to growing peptide chains on the ribosome, HSPs prevent their nonspecific aggregation, protect against premature proteolytic degradation, and promote the correct and timely folding of the polypeptide into a tertiary structure. HSPs also bind altered proteins or proteins whose tertiary structure has already formed incorrectly, protecting the cell from their effects [7]. When exposed to stressful factors, HSP activity increases dramatically. They intensively bind to denatured proteins and maintain them in a state capable of subsequent recovery. HSPs are present in the cytoplasm in combination with a special transcription factor HSF (from the English heat shock factor – heat shock factor). Under stress, HSF separates from HSP, acquires DNA binding activity and accumulates in the nucleus, where it activates transcription of new chaperones and suppresses transcription of other genes. At the end of stress exposure, the released HSPs bind HSF and return to their initial state [8-10]. By migrating to the nucleus and binding to chromatin and the nucleolus, HSPs thereby prevent the appearance of mutations and provide conditions for repairing DNA damage [11-14]. Interacting with microtubules and microfilaments, HSPs stabilize the cytoskeleton, which increases the cell's resistance to mechanical damage, denaturation, and aggregation of cell proteins. HSP70 is a family of heat shock proteins with a molecular weight of about 70 kDa, the most widespread [7]. The aim was to evaluate the content of HSP70 in the brain of rats with cerebral ischemia of varying severity.
Methods.
The experiments were performed on 24 male mongrel white rats weighing 260 ±20 g in compliance with the requirements of Directive 2010/63/EU of the European Parliament and of the Council of 22.09.2010 on the protection of animals used for scientific purposes. Cerebral ischemia was modeled under conditions of intravenous thiopental anesthesia (40-50 mg/kg). Models of partial (CIGM), stepwise subtotal (CIGM), subtotal (SIGMA) and total (TIGM) cerebral ischemia were used in the studies [2]. Partial cerebral ischemia (CHIGM) was modeled by ligation of one common carotid artery (CCA) on the right. Stepwise subtotal IGM (SSIGM) was performed by ligating both OCAs at intervals of 7 days (subgroup 1), 3 days (subgroup 2) or 1 day (subgroup 3) Subtotal cerebral ischemia (SIGMA) was modeled by simultaneous ligation of both OCAs. Total cerebral ischemia (TIGM) was modeled by animal decapitation. Brain sampling was performed after 1 hour to determine the HSP70 content. The HSP70 content was determined by the immunohistochemical method using monoclonal antibodies. For this purpose, after decapitation, the brain was quickly extracted from rats, pieces of the cerebral cortex were fixed in zinc-ethanol-formaldehyde at +4 ° C (overnight), and then enclosed in paraffin. Paraffin sections with a thickness of 5 microns were prepared using a microtome and mounted on slides. The preparations were processed according to the protocol of the immunocytochemical reaction for light International Journal of Clinical Reports and Studies microscopy, which excludes the procedure of thermal unmasking of antigens. To determine the immunoreactivity of the molecular marker HIF, primary polyclonal rabbit antibodies Anti- HSP70 antibody from Abcam (Great Britain, ab 181606) were used at a dilution of 1:1000 at +4 ° C, exposure 20 hours, in a humid chamber [4]. The EXPOSE Mouse and Rabbit specific HRP/DAB detection IHC kit Abcam (Great Britain, ab. 80436) was used to detect bound primary antibodies. The Anti-HSP70 content was studied in the cytoplasm of neurons of the fifth layer of the parietal cortex and neurons of the CA1 field of the hippocampus in immunohistochemical preparations based on the optical density of the chromogen precipitate using an Axioscop 2 plus microscope (Zeiss, Germany), a digital video camera (LeicaDFC 320, Germany) and the ImageWarp image analysis program (Bitflow, USA). Groups Temporal Cortex Control 0.161 (0.158; 0.163) 0.152 (0.148; 0.159) PIOTB 0.160 (0.159; 0.162) SSIOTB – subgroup 1 0.214 (0.183; 0.248) * 0.151 (0.146; 0.159) 0.173 (0.161; 0.182) SSIOTB – subgroup 2 0.158 (0.156; 0.162) SSIOTB – subgroup 3 0.148 (0.148; 0.160) 0.139 (0.124; 0.152) 0.134 (0.117; 0.144) SIOFB TCIOTB 0.114 (0.113; 0.117) * 0.105 (0.093; 0.119) * Page 2 of 5 To prevent systematic measurement errors, brain samples from the compared control and experimental groups of animals were studied under the same conditions. As a result of the research, quantitative continuous data has been obtained. Since the experiment used small samples with an abnormal distribution, the analysis was performed using nonparametric statistical methods using the licensed computer program Statistica 10.0 for Windows (StatSoft, Inc., USA). The data is presented as Me (LQ; UQ), where Me is the median, LQ is the value of the lower quartile, and UQ is the value of the upper quartile. The differences between the groups were considered significant at p<0>
Results
There was no change in the HSP70 content in rats with CHIGM compared to the level in the control group (p>0.05) (Table 1, Figures 1, 2).
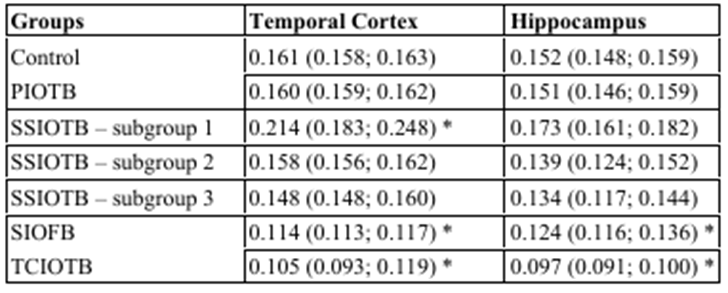
Table 1: The content of HSP70 in the cytoplasm of pyramidal neurons of the parietal cortex and field CA1 of the hippocampus of the brain of rats with cerebral ischemia, Me (LQ; UQ)Notes:
PIOTB – partial ischemia of the brain
SSIOTB – subtotal stepwise ischemia of the brain
SIOFB – subtotal ischemia of the brain
TCIOTB – total cerebral ischemia
Note: * – p<0>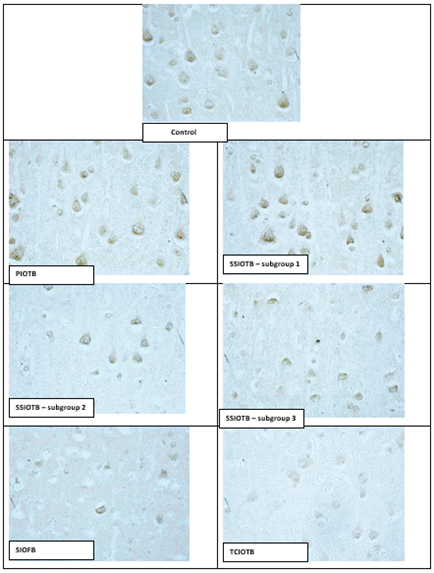
Digital micrography. Increased. 40
Figure 1: HSP70 content in the cytoplasm of pyramidal neurons of the parietal cortex of rats with partial (PIOTB), stepwise subtotal (SSIOTB), subtotal (SIOFB) and total (TCIOTB) cerebral ischemia
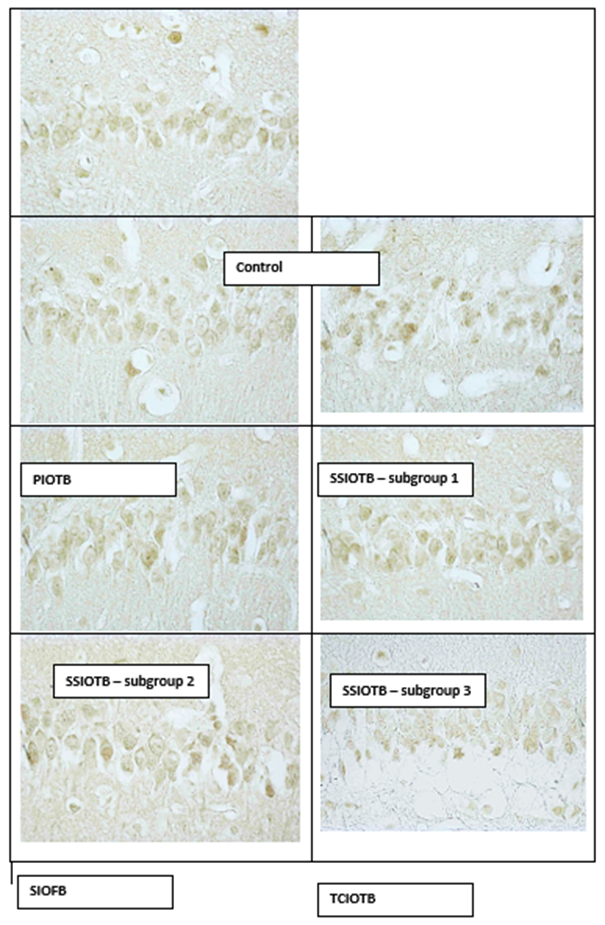
Digital micrography. Increased.
Figure 2: The content of HSP70 in the cytoplasm of pyramidal neurons of the CA1 hippocampal field of rats with partial (PIOTB), stepwise subtotal (SSIOTB), subtotal (SIOFB) and total (TCIOTB) cerebral ischemia.
Compared with the "control" group, in the 1st subgroup of SSGM (the interval between dressings was 7 days), there was a 25% increase in HSP70 content in the parietal cortex (p<0>0.05). In the 2nd and 3rd subgroups of the CGM (the interval between dressings of 3 days and 1 day), the HSP70 content did not change in any of the studied departments compared to the control group (p>0.05), and compared with the 1st subgroup, it was 26% lower in the 2nd subgroup. in the parietal cortex (p<0>0.05).
Thus, in the 1st subgroup of SSGMS with an interval between dressings of 7 days, an increase in the content of HSP70 was observed, while in the 2nd and 3rd subgroups this indicator did not differ from the level of the control group.
Compared with the control, in the SIGMA group modeled by simultaneous ligation of both OCAs, the HSP70 content decreased by 29% in the parietal cortex (p<0>
In total cerebral ischemia, the most significant decrease in HSP70 content was noted, compared with the control – by 35% in the parietal cortex, p<0>
A decrease in HSP70 was noted in subtotal total cerebral ischemia as a reflection of protein degradation. The synthesis of heat shock proteins is a universal response to stress and plays an important role in protecting cells from negative effects. Heat shock proteins are highly involved in the implementation of fundamental cellular processes, and changes in their expression can serve as an important diagnostic marker of a cell's response to damage [7-14]. The search for medicinal substances acting as inducers or inhibitors of their synthesis is an urgent area in experimental pharmacology, since it allows not only to regulate the processes of adaptation to hypoxia, but also to treat cerebrovascular, cardiovascular and other diseases more effectively, in the genesis of which oxygen deficiency plays a leading role.
References
- Belenichev IF, (2014). Neuroprotection and neuroplasticity: monograph, OOO “Polygraph Plus”, Kyiv, 512 p.
View at Publisher | View at Google Scholar - Bon EI, Maksimovich NE, (2018). Modeling methods and morphofunctional markers of cerebral ischemia, Biomedicine, (2), 59–71.
View at Publisher | View at Google Scholar - Bon EI, Maksimovich NE, (2021). Comparative analysis of morphological disorders of neurons of the parietal cortex and hippocampus of rats in various types of experimental cerebral ischemia, Orenburg Medical Bulletin, (2), 29–36.
View at Publisher | View at Google Scholar - Korzhevsky DE, Gilerovich EG, Kirik OV, (2016). Immunohistochemical study of the brain, SpetsLit, St. Petersburg, 143 p.
View at Publisher | View at Google Scholar - Maksimovich NE, Bon EI, Zimatkin SM, (2020). Rat brain and its reaction to ischemia: monograph, GrSMU, Grodno, 240 p.
View at Publisher | View at Google Scholar - Clemens JA, (2000). Cerebral ischemia: gene activation, neuronal injury, and the protective role of antioxidants, Free Radical Biology and Medicine, 28, 1526–1531.
View at Publisher | View at Google Scholar - Maksimovich NE, Bon EI, (2020). Proteins of heat shock: features and role in adaptation, methodological approaches to the definition, Biomedicine, (2), 60–67.
View at Publisher | View at Google Scholar - Maksimovich NYe, Bon IL, (2023). The role of heat shock proteins in cell metabolism, Journal of Medical and Clinical Case Studies, 1(1), 1–8.
View at Publisher | View at Google Scholar - Cui Y, Wang M, Yin X, Xu G, Song S, Li M, Liu K, Xia X, (2019). OsMSR3, a small heat shock protein, confers enhanced tolerance to copper stress in Arabidopsis thaliana, International Journal of Molecular Sciences, 20, 3–23.
View at Publisher | View at Google Scholar - Fabczak H, Osinka A, (2019). Role of the novel Hsp90 co-chaperones in dynein arms' preassembly, International Journal of Molecular Sciences, 20, 24–29.
View at Publisher | View at Google Scholar - Gupta A, Bansal A, Hashimoto-Torii K, (2020). HSP70 and HSP90 in neurodegenerative diseases, Neuroscience Letters, 716, 1–5.
View at Publisher | View at Google Scholar - Min HJ, Choe JW, Chang MY, Kim KS, Lee SY, Mun SK, (2017). The expression and correlation of Hsp70 and Hsp27 in serous middle ear effusion fluids of pediatric patients: a preliminary study, International Journal of Pediatric Otorhinolaryngology, 101, 145–149.
View at Publisher | View at Google Scholar - Mo ZT, Li WN, Zhai YR, Gao SY, (2017). The effects of icariin on the expression of HIF-1α, HSP-60 and HSP-70 in PC12 cells subjected to oxygen-glucose deprivation-induced injury, Pharmacology & Biology, 55, 848–852.
View at Publisher | View at Google Scholar - Oh E, Lee B, Choi YM, (2019). Associations of heat-shock protein expression with meat quality and sensory quality characteristics in highly marbled Longissimus thoracis muscle from Hanwoo steers categorized by Warner-Bratzler shear force value, Foods, 8, 12–18.
View at Publisher | View at Google Scholar

 Clinic
Clinic
