Research Article | DOI: https://doi.org/DOI:10.31579/2835-2971/065
ZnT8A as an Autoantibody Potential Marker for Clinical Applications in The Type 1 Diabetic Newly Diagnosed Children.
- Mona Eid Ahmed 1
- Ahmad Mohamed Awad 2
- Khaled Hassaan Awad 3
- Amr Moustafa Abdelalim Khalifa 4
- Nesma Mohammed Bahaa Eldeen 5
- M. Ashraf A. Ali 6
- Haytham Ali 6,7
- Mohamed Elhabet 8
- Hassnaa Ibrahim Montaser 9
- Samer Alsayed 10
- Mohamed Zaeim Hafez 11*
- Ahmed G. Hamad 12
1National Nutrition Institute, Cairo, Qasr El Aini St., 11562, Egypt.
2Medical biochemistry department, Faculty of Medicine, Al-Azhar University, Cairo, Egypt.
3Pediatric department, Faculty of Medicine, Al-Azhar University (Assiut), Assiut, Egypt.
4Shadadia Clinic, Farwaniyah governorate, Primary Health Care, MOH- Kuwait.
5Department of Biochemistry, Faculty of Medicine, Cairo University, Cairo, Egypt.
6Nursing department, AlGhad College for Applied Medical Sciences in Najran, KSA.
7Physiology department, Qena faculty of medicine, South valley university, Egypt.
8Surgery specialist, Department of medicine, Vision Colleges, KSA.
9Pediatrics specialist, MSc., degree Mansoura University, Mansoura, Egypt.
10Pediatrics specialist, MSc., degree Ain shams University, Cairo, Egypt.
11Medical physiology department, Faculty of Medicine, Al-Azhar University (Assiut), Assiut, Egypt.
12Anatomy and Embryology Department, Faculty of Medicine, Mansoura University, 35516, Egypt.
*Corresponding Author: Mohamed Zaeim Hafez, Medical physiology department, Faculty of Medicine, Al-Azhar University (Assiut), Assiut, Egypt.
Citation: Mona Eid Ahmed, Ahmad Mohamed Awad, Khaled Hassaan Awad, Amr Moustafa Abdelalim Khalifa, Nesma Mohammed Bahaa Eldeen, et al., (2026), ZnT8A as an Autoantibody Potential Marker for Clinical Applications in The Type 1 Diabetic Newly Diagnosed Children, J. Clinical Pediatrics and Mother Health, 5(1); DOI:10.31579/2835-2971/065
Copyright: © 2026, Mohamed Zaeim Hafez. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 05 May 2026 | Accepted: 10 May 2026 | Published: 20 May 2026
Keywords: Type 1 diabetes mellitus; children; zinc transporter 8 autoantibody (ZnT8a); autoimmune diabetes; biomarkers; early diagnosis; roc curve analysis
Abstract
Background: Type 1 diabetes is an autoimmune illness that destroys pancreatic β-cells. Early diagnosis is based on islet autoantibodies; however, traditional markers such as GAD65 and IA-2 do not detect all individuals. Zinc transporter 8 autoantibody (ZnT8A) has emerged as a promising biomarker that may increase diagnosis accuracy in newly diagnosed children with T1DM. This work aims to assess the role of ZnT8A as an autoantibody potential marker for clinical applications in the type 1 diabetic newly diagnosed children.
Methods: This case-control study included newly diagnosed children with T1DM as well as healthy controls of similar age and gender. Clinical evaluation and laboratory investigations were carried out, including fasting blood glucose, HbA1c, serum insulin, C-peptide, and standard biochemical assays. An enzyme-linked immunosorbent test (ELISA) was used to assess islet autoantibodies (ZnT8A, GAD65-Ab, IA-2-Ab, and insulin autoantibodies). Statistical analysis was carried out with appropriate comparative tests, and the diagnostic performance of ZnT8A was evaluated utilizing receiver operating characteristic (ROC) curve analysis.
Results: ZnT8A levels were substantially higher in newly diagnosed T1DM youngsters than in healthy controls. The antibody showed excellent diagnostic performance, with high sensitivity and specificity in discriminating T1DM patients from controls. ZnT8A positive was common even in individuals who tested negative for other islet autoantibodies, which improved overall diagnostic yield when paired with GAD65 and IA-2 antibodies. In addition, diabetic children had significantly higher HbA1c and lower C-peptide and serum insulin levels than controls.
Conclusion: ZnT8A is a very useful autoimmune biomarker in newly diagnosed Type 1 diabetic children. Its inclusion in regular autoantibody panels improves diagnostic sensitivity and early detection of autoimmune diabetes. ZnT8A may be a clinically relevant marker for early identification and illness categorization in pediatric T1DM.
Graphical Abstract
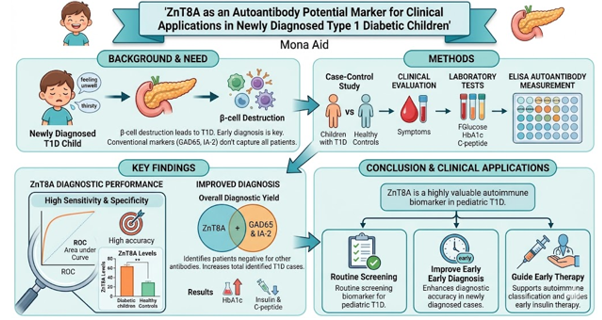
The graphical abstract visually summarizes a case-control study evaluating ZnT8A as a novel autoantibody biomarker for early detection of Type 1 Diabetes (T1D) in newly diagnosed pediatric patients. The illustration is structured into four integrated sections: [1]. the clinical context, depicting B-cell destruction as the pathogenic driver of T1D and highlighting limitations of conventional markers (GAD65, IA-2) in capturing all cases; [2]. methodological workflow, showing parallel evaluation of newly diagnosed T1D children versus healthy controls through clinical symptom assessment (e.g., fatigue, polydipsia) and laboratory measurements (fasting glucose, HbA1c, C-peptide, and ZnT8A via ELISA); [3]. key diagnostic findings, represented by a ROC curve demonstrating ZnT8A's high sensitivity and specificity, alongside its capacity to identify seronegative patients missed by standard antibodies, thereby increasing overall diagnostic yield; and [4]. clinical translation, emphasizing how routine ZnT8A screening can enhance early diagnosis accuracy, support autoimmune subclassification, and guide timely initiation of insulin therapy. Color-coded icons, directional arrows, and concise annotations ensure rapid visual navigation, making the abstract suitable for high-impact pediatric endocrinology or diabetes journals while clearly communicating the translational value of ZnT8A as a complementary biomarker in pediatric T1D management.
Key Messages
• Zinc transporter 8 autoantibody (ZnT8A) is a highly specific indicator of autoimmune β-cell damage in newly diagnosed Type 1 diabetes in children.
• ZnT8A has excellent diagnostic performance, with high sensitivity and specificity, demonstrating its utility in separating Type 1 diabetes from non-autoimmune types of diabetes and healthy persons.
• Adding ZnT8A to autoantibody panels enhances early diagnosis accuracy for children Type 1 diabetes.
• ZnT8A positive is especially relevant in early-stage or freshly diagnosed patients, where other clinical or biochemical indicators may remain equivocal.
• Detection of ZnT8A confirms the autoimmune character of diabetes, directing early insulin therapy and disease classification.
Introduction
Type 1 diabetes mellitus (T1DM) represents one of the most prevalent chronic metabolic conditions affecting the pediatric population. The disease arises from autoimmune-mediated destruction of pancreatic β-cells, leading to absolute insulin deficiency and necessitating lifelong exogenous insulin replacement. The primary therapeutic objective is to maintain glycemic control within recommended targets, typically monitored via hemoglobin A1c (HbA1c) assessment (Addissouky et al., 2024). Risk factors for T1DM encompass endogenous, exogenous, and non-modifiable variables that collectively increase susceptibility in specific pediatric subgroups and inform strategies for primary prevention (Mohammed et al., 2024). Clinical diagnosis of diabetes is established when classic hyperglycemic symptoms such as polyuria, polydipsia, unexplained weight loss, polyphagia, fatigue, or blurred vision secondary to osmotic lens changes are accompanied by a random plasma glucose concentration ≥200 mg/dL (11.1 mmol/L). These manifestations may persist for days to weeks prior to recognition. In young children, particularly toddlers and prepubertal girls, perineal candidiasis frequently serves as an early clinical indicator (Chiang et al., 2018). Management of pediatric T1D remains challenging, especially among preschool-aged children and adolescents. In many regions, mean or median HbA1c values consistently exceed the recommended threshold of 7.5%, reflecting suboptimal glycemic control (Hatun et al., 2024). Zinc transporter 8 autoantibodies (ZnT8A) have been identified as one of four principal islet-directed autoantibodies, alongside glutamic acid decarboxylase 65 (GADA), islet antigen-2 (IA-2A), and insulin autoantibodies (IAA) (Bhola et al., 2021). The SLC30A8 gene encodes ZnT8, and genome-wide association studies have mapped a susceptibility locus to chromosome 8q24.11. A common single-nucleotide polymorphism, rs13266634 (C/T), results in an arginine-to-tryptophan substitution at position 325 (R325W) within the protein sequence (Williams & Long, 2019). ZnT8 autoantibodies hold significant promise as a diagnostic adjunct in T1DM. ZnT8 is a polytopic transmembrane protein comprising 369 amino acids, with cytoplasmic N- and C-terminal domains, predominantly localized to insulin secretory granules within pancreatic β-cells (Williams & Long, 2019; Huang et al., 2019). As the most abundantly expressed zinc transporter in β-cells, ZnT8 facilitates zinc ion (Zn²⁺) transport essential for insulin crystallization, storage, and regulated secretion (De Oliveira et al., 2024). Autoantibodies targeting ZnT8 may interfere with insulin biosynthesis, granule maturation, exocytosis, and intra-islet paracrine/autocrine signaling (Khaleel et al., 2025). Notably, ZnT8-directed autoimmunity often emerges years before clinical symptom onset, and rising ZnT8A titers correlate with accelerated β-cell loss, positioning this biomarker as a potential indicator of disease progression and activity (Fuentes-Cantero et al., 2024). Given its high β-cell specificity and functional centrality in insulin physiology, ZnT8 represents both a mechanistically relevant autoantigen and a valuable tool for early detection and risk stratification in pediatric T1DM. We hypothesize that Zinc Transporter 8 Autoantibody (ZnT8A) functions as a highly sensitive, specific, and clinically complementary biomarker for the early diagnosis and autoimmune subclassification of Type 1 Diabetes Mellitus (T1DM) in newly diagnosed pediatric patients. This study aims to, a) to quantify and compare circulating ZnT8A levels between newly diagnosed T1DM children and age- and sex-matched healthy controls, b) To assess the diagnostic performance of ZnT8A using sensitivity, specificity, predictive values, and receiver operating characteristic (ROC) curve analysis, c) To determine the additive diagnostic yield of ZnT8A when integrated with conventional islet autoantibody panels (GAD65, IA-2, and IAA), with emphasis on identifying seronegative cases missed by standard markers, and d) To correlate ZnT8A positivity and titers with key clinical and biochemical parameters, including HbA1c, fasting blood glucose, serum insulin, and C-peptide, as surrogate indicators of residual β-cell function and glycemic severity.
Patients And Methods
Study Design and Setting
This case-control investigation was conducted to evaluate the diagnostic utility of Zinc Transporter 8 Autoantibody (ZnT8A) as a potential clinical biomarker in pediatric patients with newly diagnosed Type 1 Diabetes Mellitus (T1DM). The study population comprised two age- and sex-matched cohorts: Group I (Cases) included children meeting the American Diabetes Association (ADA) diagnostic criteria for T1DM characterized by hyperglycemia, insulin dependence, and clinical features consistent with autoimmune β-cell destruction; Group II (Controls) consisted of healthy children without personal or family history of diabetes or other autoimmune disorders.
Eligibility Criteria
Inclusion criteria: Participants aged 1–18 years with a confirmed T1DM diagnosis within the preceding 3 months and no prior exposure to prolonged insulin therapy (>2 weeks before sample collection).
Exclusion criteria: Presence of chronic hepatic, renal, or systemic inflammatory conditions; diagnosis of Type 2 diabetes, monogenic diabetes, or other forms of non-autoimmune diabetes; active or recent infection within 4 weeks; and current or recent use of immunosuppressive or immunomodulatory medications.
Clinical and Laboratory Assessments
All enrolled subjects underwent comprehensive clinical evaluation, including collection of demographic information (age, sex), anthropometric measurements (weight, height, body mass index), and documentation of presenting symptoms (polyuria, polydipsia, unexplained weight loss, diabetic ketoacidosis).
Routine Biochemical Profiling: Following an overnight fast, venous blood samples were obtained for standardized laboratory analyses:
Complete Blood Count (CBC): Automated hematology analysis provided hemoglobin concentration (g/dL), total and differential leukocyte counts (×10³/µL), and platelet count (×10³/µL) to screen for anemia, infection, or hematological abnormalities.
Glycemic Parameters: Fasting blood glucose (FBG) was quantified using the glucose oxidase-peroxidase enzymatic method (mg/dL). Glycated hemoglobin (HbA1c) was measured by high-performance liquid chromatography (HPLC) aligned with NGSP standards and reported as a percentage, reflecting mean glycemia over the preceding 2–3 months.
β-Cell Function Markers: Fasting serum insulin (µIU/mL) was assessed via chemiluminescent immunoassay (CLIA), while C-peptide (ng/mL) was determined by enzyme-linked immunosorbent assay (ELISA) to evaluate residual endogenous insulin secretion.
Hepatic and Renal Function: Liver enzymes (ALT, AST; U/L) were measured using kinetic UV-IFCC methodology; serum albumin (g/dL) was quantified by the bromocresol green method. Renal function was assessed via serum urea (urease enzymatic method) and creatinine (Jaffe kinetic method), both expressed in mg/dL.
Autoimmune Serology: ZnT8A levels were quantified in serum using a validated ELISA platform per manufacturer protocols. Results were expressed in IU/mL, with positivity defined according to assay-specific cutoff thresholds. All laboratory procedures were performed in a central accredited laboratory under rigorous internal and external quality assurance protocols.
Ethical considerations status
The study protocol adhered to the ethical principles outlined in the Declaration of Helsinki, guidelines from the Islamic Organization for Medical Sciences, World Health Organization standards, and International Council for Harmonisation Good Clinical Practice. Ethical approval was granted by the Research Ethics Committee, Faculty of Medicine, Al-Azhar University (Assiut) (Reference: RESEARCH/AZ.AST./PHS006/6/252/5/2025). Written informed consent was obtained from parents or legal guardians following comprehensive explanation of study objectives, procedures, potential benefits, and risks. Participant assent was sought where age-appropriate.
Sample size estimation
Sample size calculation was based on a case-control design to detect a significant difference in ZnT8A seropositivity between T1DM cases and healthy controls. Assuming a two-sided α of 0.05, 80% statistical power (β = 0.20), and anticipated ZnT8A positivity rates of ~70% in cases versus ~5% in controls (derived from prior literature), the minimum required sample was estimated at approximately 35 participants per group using standard formulas for comparing two independent proportions. To enhance robustness and accommodate potential attrition or incomplete datasets, the target sample was increased by 10–20%, ensuring adequate power for detecting clinically meaningful differences in ZnT8A levels between groups.
Statistical analysis methods
Data management and statistical analyses were performed using IBM SPSS Statistics for Windows (Version 25.0; IBM Corp., Armonk, NY, USA) and MedCalc Statistical Software (Version 19.6.1; MedCalc Software Ltd., Ostend, Belgium). Qualitative variables were summarized as frequencies and percentages; quantitative variables were expressed as mean ± standard deviation (SD), median, and interquartile range, as appropriate. Normality of distribution was assessed using the Shapiro-Wilk test. Between-group comparisons employed the Chi-square (χ²) test for categorical variables, Mann-Whitney U test for non-normally distributed continuous variables, and Kruskal-Wallis H test for multi-group comparisons. Diagnostic performance of ZnT8A was evaluated using receiver operating characteristic (ROC) curve analysis, with area under the curve (AUC), sensitivity, specificity, and predictive values calculated. Heatmap plot correlation was used to assess the correlation between all variables under study. A two-tailed p-value <0>
Results
Study Population and Participant Flow
A total of 113 children initially diagnosed with Type 1 Diabetes Mellitus (T1DM) at the Faculty of Medicine were screened for eligibility. Thirteen participants were excluded (five declined consent; eight did not meet inclusion criteria), yielding a final cohort of 100 children stratified into two groups: newly diagnosed T1DM cases and age- and sex-matched healthy controls (Figure 1).
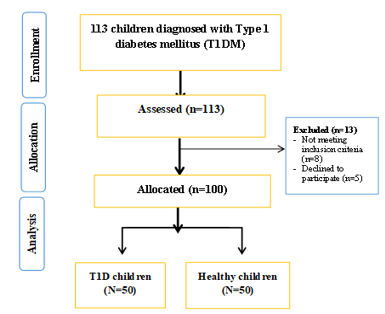
Figure 1: Flowchart of the studied groups
Demographic and clinical characteristics
No statistically significant differences were observed between groups regarding age, sex, body mass index (BMI), or consumption of high-glycemic-index foods. However, significant between-group differences were noted for mode of delivery (cesarean section) and family history of T1D. Among the T1DM cohort, mean age at diagnosis and mean disease duration were 6.38 ± 2.25 years and 4.84 ± 2.10 years, respectively (Table 1).
T1D children (N=50) | Healthy children (N=50) | P value | |
| Age/year | 7.70±2.63 | 8.00±5.66 | 0.879 |
Sex Females Males | 20 (40.0) 30 (60.0) | 25 (50.0) 25 (50.0) | 0.315 |
| Cesarean delivery | 25 (50.0) | 35 (70.0) | 0.041* |
| Body mass index (kg/m2) | 17.10±6.14 | 16.94±6.19 | 0.897 |
| High-glycemic index food intake | 36 (72.0) | 28 (56.0) | 0.096 |
Family History of T1D Father Mother Sibling Multiple relatives | 28 (56.0) 11 (22.0) 9 (18.0) 2 (4.0) | 0 (0.0) 0 (0.0) 0 (0.0) 0 (0.0) | <0> |
| Age at diagnosis (yr) | 6.38±2.25 | ---± --- | --- |
| Duration of diabetes (yr) | 4.84±2.10 | ---± --- | --- |
Table 1: Demographic and clinical data among the studied groups.
Biochemical and Immunological Profiles
Compared with healthy controls, children with T1DM exhibited significantly elevated levels of white blood cell count (WBC), platelets, urea, alanine aminotransferase (ALT), aspartate aminotransferase (AST), glycated hemoglobin (HbA1c), thyroid-stimulating hormone (TSH), and Zinc Transporter 8 Autoantibody (ZnT8A). Conversely, hemoglobin, creatinine, albumin, fasting serum insulin, C-peptide, and free triiodothyronine (Free T3) were significantly reduced in the diabetic group. ZnT8A seropositivity also differed significantly between groups. No significant intergroup differences were detected for creatinine or free thyroxine (Free T4) (Table 2, Figure 2).
T1D children (N=50) | Healthy children (N=50) | P value | |
| Hemoglobin (Hb), g/dl | 11.55±0.90 | 12.64±0.70 | <0> |
| WBCS *1000/µl | 7.64±1.58 | 7.08±1.14 | 0.044* |
| Platelet *1000/µl | 297.90±35.07 | 281.20±37.78 | 0.024* |
| Urea (mg/dl) | 29.77±5.03 | 25.43±4.61 | <0> |
| Creatinine (mg/dl) | 0.68±0.15 | 0.64±0.12 | 0.107 |
| ALT (U/L) | 32.42±7.24 | 25.86±6.07 | <0> |
| AST (U/L) | 33.00±6.20 | 24.62±5.97 | <0> |
| Albumin (g/dl) | 4.15±0.26 | 4.32±0.18 | <0> |
| HbA1c (%) | 10.70±1.43 | 5.12±0.25 | <0> |
| ZnT8A-positive | 36 (72.0) | 0 (0.0) | <0> |
| ZnT8A-negative | 15 (30.0) | 0 (0.0) | <0> |
Table 2: Laboratory investigations among the studied groups.
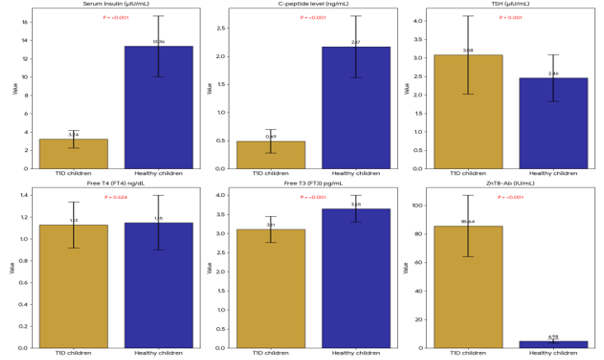
Figure 2: Serum Insulin, C-peptide level, thyroid hormones and ZnT8-Ab levels among the studied child.
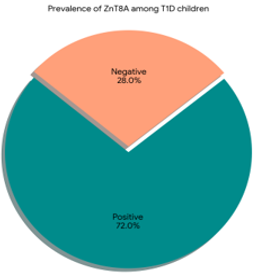
Figure 3: ZnT8-Ab levels among the studied T1D children. In the current study, there was 36 T1 diabetic children had ZnT8A-positive (n=3672.0%) and 15 patients had ZnT8A-negative (n=15, 30.0%).
Stratification by ZnT8A Serostatus within the T1DM Cohort
Among children with T1DM, ZnT8A seropositivity was not significantly associated with age, sex, BMI, dietary glycemic index, age at diagnosis, disease duration, or most hematological, hepatic, renal, or endocrine parameters. However, cesarean delivery was significantly associated with ZnT8A status. Additionally, ZnT8A-negative children demonstrated significantly higher urea and HbA1c levels compared with ZnT8A-positive counterparts (Table 3, Figure 4).
T1D children | ZnT8A-positive (N=36) | ZnT8A-negative (N=14) | P value |
| Age/year | 7.25±2.50 | 8.86±2.68 | 0.072 |
Sex Females Males | 16 (44.4) 20 (55.6) | 4 (28.6) 10 (71.4) | 0.304 |
| Cesarean delivery | 14 (38.9) | 11 (78.6) | 0.012* |
| Body mass index (kg/m2) | 16.17±6.05 | 19.50±5.89 | 0.148 |
| High-glycemic index food intake | 24 (66.7) | 12 (85.7) | 0.178 |
| Age at diagnosis (yr) | 6.47±2.26 | 6.14±2.28 | 0.616 |
| Duration of diabetes (yr) | 4.89±2.04 | 4.71±2.33 | 0.870 |
| WBCS *1000/µl | 7.35±1.55 | 8.37±1.46 | 0.050 |
| Platelet *1000/µl | 294.17±35.87 | 307.50±32.15 | 0.420 |
| Urea (mg/dl) | 28.78±5.16 | 32.32±3.76 | 0.020* |
| Creatinine (mg/dl) | 0.66±0.14 | 0.73±0.15 | 0.203 |
| ALT (U/L) | 32.50±7.09 | 32.21±7.90 | 0.957 |
| AST (U/L) | 33.12±6.68 | 32.69±4.96 | 0.648 |
| Albumin (g/dl) | 4.13±0.27 | 4.19±0.25 | 0.417 |
| HbA1c (%) | 10.48±1.44 | 11.26±1.26 | 0.023* |
Table 3: Demographic, clinical data and Laboratory investigations in relation to positive and negative ZnT8A among T1D children.
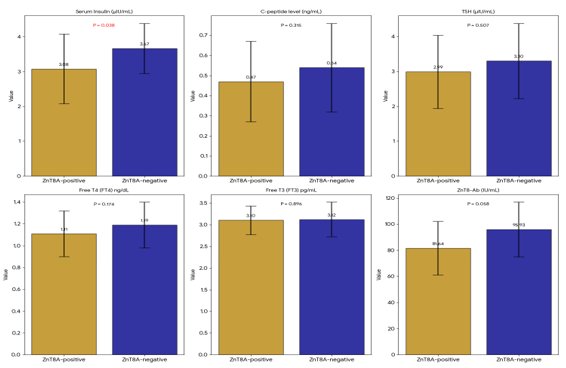
Figure 4: Serum Insulin, C-peptide level, thyroid hormones and ZnT8-Ab levels among T1D children with positive and negative ZnT8A.
Diagnostic Performance of ZnT8A: ROC Curve Analysis
Receiver operating characteristic (ROC) analysis identified an optimal ZnT8A cutoff of >52 IU/mL for discriminating newly diagnosed T1DM children from healthy controls, yielding a sensitivity of 98.7%, specificity of 79.2%, and area under the curve (AUC) of 0.968 (Figure 5a). When comparing ZnT8A-positive versus ZnT8A-negative children within the T1DM cohort, a higher cutoff (>74 IU/mL) was identified, with sensitivity of 97.3%, specificity of 72.3%, and AUC of 0.673 (Figure 5b), indicating stronger diagnostic utility for case-control discrimination than for intra-cohort stratification.
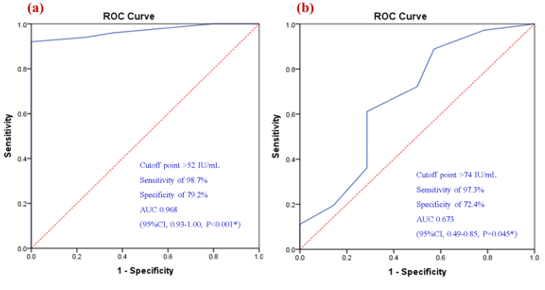
Figure 5: Roc curve analysis of ZnT8-Ab levels for a) early detection of T1D children compared to control children and b) early detection of T1D children among ZnT8A-positive compared to ZnT8A-negative children.
Correlation Analyses
Significant positive correlations were observed between serum insulin and HbA1c, and a significant negative correlation with BMI; no other significant insulin correlations were detected. C-peptide levels correlated positively with disease duration, hemoglobin, WBC, platelets, albumin, and HbA1c. Free T3 showed positive correlations with disease duration, hemoglobin, WBC, urea, albumin, and C-peptide, and a negative correlation with age at diagnosis. Free T4 correlated positively with BMI, age at diagnosis, urea, AST, HbA1c, and albumin, and negatively with disease duration. No other significant correlations were identified for these markers (Figure 6).
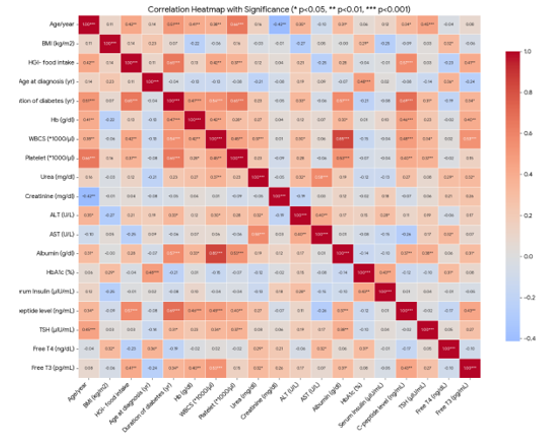
Figure 6: Heatmap plot correlation matrix between ZnT8A-levels and all of the studied variables.
Discussion
Type 1 Diabetes Mellitus represents a chronic autoimmune condition characterized by immune-mediated destruction of pancreatic β-cells, resulting in absolute insulin deficiency and lifelong dependence on exogenous insulin therapy (Azim, 2026). Prompt and accurate diagnosis in pediatric populations is critical to prevent acute metabolic complications and to facilitate precise disease subclassification (Chiang et al., 2018). Autoantibodies targeting pancreatic islet antigens serve as essential diagnostic tools, with Zinc Transporter 8 Autoantibody (ZnT8A) emerging as a promising biomarker following the identification of ZnT8 as a major T1D autoantigen (Wenzlau et al., 2007, 2009). Accumulating evidence indicates that integrating ZnT8A into conventional autoantibody panels—comprising GAD65, IA-2, and insulin autoantibodies enhances diagnostic sensitivity, particularly among newly diagnosed children and individuals previously classified as autoantibody-negative (Lampasona et al., 2010; Fakhfakh et al., 2022). Given that ZnT8A prevalence and performance may vary by age, ethnicity, and assay methodology, population-specific validation remains essential (Niechciał et al., 2018; Rochmah et al., 2020). Consequently, evaluating ZnT8A as a clinical marker in newly diagnosed pediatric T1DM may strengthen early detection and refine autoimmune classification strategies. In the present study, children with newly diagnosed T1DM exhibited significantly elevated WBC, platelets, urea, hepatic transaminases (ALT, AST), HbA1c, TSH, and ZnT8A levels relative to healthy controls, alongside reduced hemoglobin, albumin, fasting insulin, C-peptide, creatinine, and Free T3. These alterations reflect the characteristic metabolic dysregulation, autoimmune activation, and partial β-cell loss accompanying new-onset disease. Critically, ZnT8A was markedly elevated in diabetic children, reinforcing its utility as a clinically relevant autoimmune marker for pediatric T1DM diagnosis. Our findings align with foundational work by Wenzlau et al. (2007, 2009), who first characterized ZnT8 as a major T1D autoantigen, and subsequent studies confirming that ZnT8A inclusion improves diagnostic sensitivity beyond conventional antibodies alone. Similarly, Fakhfakh et al. (2022) reported that ZnT8A was the sole detectable autoantibody in a subset of newly diagnosed children previously deemed autoantibody-negative, underscoring its additive diagnostic value. This concordance supports our observation that ZnT8A effectively discriminates diabetic children from healthy peers, affirming its role as a complementary marker for early autoimmune diabetes identification. The clinical relevance of ZnT8A in our cohort is further corroborated by international pediatric studies. Niechciał et al. (2018) documented substantially higher ZnT8A prevalence in children versus adults, suggesting heightened relevance in childhood-onset disease. Likewise, Głowińska-Olszewska et al. (2016) and Fabris et al. (2015) emphasized ZnT8A's high sensitivity in pediatric populations and its utility in early screening panels alongside GAD65 and IA-2 antibodies. These agreements may reflect the more vigorous autoimmune activity and robust humoral responses typically observed in children compared with adults. Nevertheless, reported ZnT8A prevalence varies considerably across ethnic groups: Fakhfakh et al. (2022) observed 42% positivity, lower than Caucasian cohorts (60–80%; Wenzlau et al., 2007) but higher than Asian populations such as Chinese and Japanese cohorts (Yang et al., 2010; Kawasaki et al., 2008). Such discrepancies likely stem from ethnic and genetic heterogeneity, HLA background variation, assay platform differences, sample size, and timing of antibody measurement. Thus, while our data support ZnT8A as a valuable diagnostic marker, population-specific considerations should guide its clinical implementation. Regarding disease duration, our study detected no significant difference in ZnT8A serostatus according to diabetes duration among newly diagnosed children, consistent with Yang et al. (2010). In contrast, Garnier et al. (2018) and Howson et al. (2012) reported higher ZnT8A prevalence proximal to disease onset with subsequent decline. This divergence may reflect our focus on a homogeneously short-duration cohort, minimizing the temporal variability observable in broader-duration populations. Within our T1DM cohort, ZnT8A positivity was not significantly associated with demographic variables (age, sex, BMI), dietary factors, age at diagnosis, or most biochemical parameters, suggesting that ZnT8A primarily reflects autoimmune status rather than metabolic or demographic heterogeneity. This aligns with evidence positioning ZnT8A as an immunological classification marker rather than a predictor of disease severity. Notably, cesarean delivery showed a significant association with ZnT8A status a finding warranting further investigation, potentially involving early-life immune programming or microbiome-mediated mechanisms. Additionally, ZnT8A-negative children exhibited higher HbA1c and urea levels than ZnT8A-positive peers, possibly indicating a subgroup with delayed autoimmune recognition, greater metabolic decompensation, or distinct pathogenic pathways. Given limited prior research directly comparing metabolic profiles by ZnT8A status, this observation merits future exploration. The elevated HbA1c observed in our cohort aligns with the established metabolic dysregulation of new-onset T1DM and concurs with El-Sayed et al. (2020), who reported markedly elevated HbA1c in pediatric T1DM patients, particularly those with concurrent hepatic dysfunction. Their study also documented significantly increased ALT and AST in children with poor glycemic control, with improvement following glycemic optimization. This supports our findings of elevated hepatic enzymes and HbA1c in T1DM children, suggesting that hyperglycemia and metabolic instability at diagnosis may contribute to transient hepatic impairment potentially via hepatocellular effects of chronic hyperglycemia, glycogenic hepatopathy, or metabolic stress. Similarly, the significantly elevated TSH and altered thyroid parameters in our diabetic cohort are supported by Omar et al. (2014), who reported increased prevalence of thyroid dysfunction, including clinical and subclinical hypothyroidism, among children with T1DM. These findings reinforce the well-established association between T1DM and autoimmune thyroid disease, likely attributable to shared autoimmune susceptibility. Collectively, these studies indicate that pediatric T1DM frequently involves multisystem biochemical and autoimmune disturbances, underscoring the importance of comprehensive endocrine and metabolic evaluation beyond glycemic assessment alone in newly diagnosed children. ROC curve analysis in our study demonstrated excellent diagnostic performance of ZnT8A for distinguishing newly diagnosed T1DM children from healthy controls, with exceptional sensitivity (98.7%), good specificity (79.2%), and an outstanding AUC of 0.968. These results strongly support ZnT8A as a highly accurate biomarker for early pediatric T1DM detection and suggest substantial clinical utility as a screening and diagnostic tool. Our findings appear to exceed several prior reports while remaining consistent in affirming ZnT8A's diagnostic value. Lampasona et al. (2011), via the Diabetes Antibody Standardization Program, reported a median ROC-AUC of 0.848 using radioligand assay supporting ZnT8A reliability but with lower overall accuracy than observed herein. Similarly, Dunseath et al. (2015) reported an AUC of 0.80 with lower sensitivity (54%) but very high specificity (99%) using ELISA, while Rochmah et al. (2020) found a moderate AUC of 0.71 with sensitivity of 73.3% and specificity of 72.2%. Collectively, these studies corroborate ZnT8A's diagnostic utility, though our markedly higher sensitivity and AUC suggest enhanced performance in our newly diagnosed pediatric cohort. In contrast, ROC analysis comparing ZnT8A-positive versus ZnT8A-negative children within the T1DM cohort yielded a lower AUC (0.673), despite high sensitivity (97.3%) and moderate specificity (72.3%). This indicates that while ZnT8A is highly effective for distinguishing T1DM patients from healthy controls, it possesses more limited discriminatory power for stratifying immunological subgroups within established T1DM. This is anticipated, as both ZnT8A-positive and ZnT8A-negative children share the same clinical diagnosis, reducing intergroup separation. Consequently, ZnT8A appears more clinically valuable as a diagnostic marker for disease presence rather than for internal disease subclassification. Overall, our ROC findings robustly reinforce ZnT8A as a highly promising biomarker for early pediatric T1DM diagnosis, with performance that may surpass prior reports in certain populations. Observed differences from existing literature likely reflect variations in age at diagnosis, disease stage, ethnicity, assay platform, and study design. These results support incorporating ZnT8A into early diagnostic panels for children, particularly in newly diagnosed cases where maximizing sensitivity is paramount.
Conclusion
This study demonstrates that Zinc Transporter 8 Autoantibody (ZnT8A) represents a highly sensitive and diagnostically accurate biomarker for early detection of Type 1 Diabetes Mellitus in newly diagnosed pediatric patients. Integration of ZnT8A into conventional autoantibody panels may enhance disease classification and reduce false-negative diagnoses. Routine clinical implementation of ZnT8A screening could facilitate earlier diagnosis, support comprehensive autoimmune evaluation, and inform timely therapeutic intervention in children with suspected T1DM.
Declarations
Data sharing statement: All the information and resources used in this work are accessible.
Consent for publication: All authors read the article, made necessary revisions, and consented to publish it; there were no conflicts of interest.
Authors contributions: Every author contributed significantly to the work reported, whether it was in the idea, study design, execution, data collection, analysis, and interpretation, or all of these areas; they all participated in the article's drafting, revision, or critical review; they all agreed on the journal to which the article was submitted; and they all agreed to take responsibility for every part of the work.
Disclosure: The authors declare no competing interest in this work.
Acknowledgements: Not applicable.
Funding: The authors have no funding source to declare
References
- Addissouky TA, Ali MM, El Sayed IE, Wang Y. (2024). Type 1 diabetes mellitus: retrospect and prospect. Bulletin of the National Research Centre. Apr 19;48(1):42.
View at Publisher | View at Google Scholar - Azim SN. Type 1 Diabetes Mellitus. (2026). InAutoimmune Diseases: Understanding, Management and Clinical Practice. Apr 1 (pp. 349-375). Cham: Springer Nature Switzerland.
View at Publisher | View at Google Scholar - Bhola S, Cave EM, Bhana S, Crowther NJ, Padoa CJ. (2021). Zinc transporter 8 (ZnT8) autoantibody prevalence in black South African participants with type 1 diabetes. BMC endocrine disorders. Jul 16;21(1):151.
View at Publisher | View at Google Scholar - Chiang JL, Maahs DM, Garvey KC, Hood KK, Laffel LM, et al. (2018). Type 1 diabetes in children and adolescents: a position statement by the American Diabetes Association. Diabetes care. Aug 13;41(9):2026.
View at Publisher | View at Google Scholar - De Oliveira Andrade LJ, de Oliveira GC, Rocha RM, Vinhaes Bittencourt AM, David TM, et al. (2024). Evaluating the Diagnostic Accuracy of Anti-Zinc Transporter 8 Antibodies in Type 1 Diabetes: A Systematic Review and Meta-Analysis. medRxiv. Jun 9:2024-2026.
View at Publisher | View at Google Scholar - Dunseath G, Ananieva-Jordanova R, Coles R, Powell M, Amoroso M, et al. (2015). Bridging-type enzyme-linked immunoassay for zinc transporter 8 autoantibody measurements in adult patients with diabetes mellitus. Clinica Chimica Acta. Jul 20;447:90-95.
View at Publisher | View at Google Scholar - El-Sayed MH, Thabet RA, Hamza MT, Hussein MS, El Saeed MM. (2020). Liver disease in children and adolescents with type 1 diabetes mellitus: A link between glycemic control and hepatopathy. Diabetes Research and Clinical Practice. Dec 1;170:108458.
View at Publisher | View at Google Scholar - Fabris M, Zago S, Liguori M, Trevisan MT, Zanatta M, et al. (2015). Anti-zinc transporter protein 8 autoantibodies significantly improve the diagnostic approach to type 1 diabetes: an Italian multicentre study on paediatric patients. Autoimmunity Highlights. Aug;6(1):17-22.
View at Publisher | View at Google Scholar - Fakhfakh R, Kmiha S, Tahri S, Feki S, Zouidi F, et al. (2022). Autoantibodies to Zinc Transporter 8 and SLC30A8 Genotype in Type 1 Diabetes Childhood: A Pioneering Study in North Africa. Journal of Diabetes Research. 2022(1):2539871.
View at Publisher | View at Google Scholar - Fuentes-Cantero S, González-Rodríguez C, Rodríguez-Chacón C, Galvan-Toribio R, Hermosín-Escudero J, et al. (2024). Study of the diagnostic efficiency of anti-ZnT8 autoantibodies for type 1 diabetes in pediatric patients. Laboratory Medicine. May 1;55(3):299-303.
View at Publisher | View at Google Scholar - Garnier L, Marchand L, Benoit M, Nicolino M, Bendelac N, et al. (2018). Screening of ZnT8 autoantibodies in the diagnosis of autoimmune diabetes in a large French cohort. Clinica Chimica Acta. Mar 1;478:162-165.
View at Publisher | View at Google Scholar - Głowińska-Olszewska B, Michalak J, Łuczyński W, del Pilar Larosa M, Chen S, et al. (2016). Organ-specific autoimmunity in relation to clinical characteristics in children with long-lasting type 1 diabetes. Journal of Pediatric Endocrinology and Metabolism. Jun 1;29(6):647-656.
View at Publisher | View at Google Scholar - Hatun Ş, Gökçe T, Can E, Eviz E, Karakuş KE, Smart C, Hanas R, Mutlu GY. (2024). Current management of type 1 diabetes in children: Guideline-based expert opinions and recommendations. Journal of clinical research in pediatric endocrinology. Sep 4.
View at Publisher | View at Google Scholar - Howson JM, Krause S, Stevens H, Smyth DJ, Wenzlau JM, et al. (2012). Genetic association of zinc transporter 8 (ZnT8) autoantibodies in type 1 diabetes cases. Diabetologia. Jul;55(7):1978-1984.
View at Publisher | View at Google Scholar - Huang Q, Du J, Merriman C, Gong Z. (2019). Genetic, functional, and immunological study of ZnT8 in diabetes. International journal of endocrinology. (1):1524905.
View at Publisher | View at Google Scholar - Kawasaki E, Uga M, Nakamura K, Kuriya G, Satoh T, Fujishima K, Ozaki M, Abiru N, Yamasaki H, Wenzlau JM, Davidson HW. (2008). Association between anti-ZnT8 autoantibody specificities and SLC30A8 Arg325Trp variant in Japanese patients with type 1 diabetes. Diabetologia. Dec;51(12):2299-2302.
View at Publisher | View at Google Scholar - Khaleel ZK, Al-Yozbakee ZM, Al-Hasso IK. (2025). Assessment of Anti-Zinc Transporter Protein-8 Function in Adults with Type 1 Diabetes. Al-Rafidain Journal of Medical Sciences (ISSN 2789-3219). Apr 18;8(2):76-81.
View at Publisher | View at Google Scholar - Lampasona V, Petrone A, Tiberti C, Capizzi M, Spoletini M,et al. (2010). Zinc transporter 8 antibodies complement GAD and IA-2 antibodies in the identification and characterization of adult-onset autoimmune diabetes: Non Insulin Requiring Autoimmune Diabetes (NIRAD) 4. Diabetes care. Jan 1;33(1):104-108.
View at Publisher | View at Google Scholar - Mohammed AG, Shaker EM, Mahmood SA. (2024). Risk Factors of Type 1 Diabetes Mellitus in Children and Adolescents: A Review. IAR Journal of Medical Sciences. June 30:1-23.
View at Publisher | View at Google Scholar - Niechciał E, Rogowicz-Frontczak A, Piłaciński S, Fichna M, Skowrońska B, et al. (2018). Autoantibodies against zinc transporter 8 are related to age and metabolic state in patients with newly diagnosed autoimmune diabetes. Acta diabetologica. Mar;55(3):287-294.
View at Publisher | View at Google Scholar - Omar MA, Rizk MM, El-Kafoury AA, Kilany D. (2014). Screening for thyroid disease among children and adolescents with type 1 diabetes mellitus. Alexandria Journal of Medicine. Mar 1;50(1):77-82.
View at Publisher | View at Google Scholar - Rochmah N, Faizi M, Windarti SW. (2020). Zinc transporter 8 autoantibody in the diagnosis of type 1 diabetes in children. Clinical and experimental pediatrics. Oct 6;63(10):402.
View at Publisher | View at Google Scholar - Wenzlau JM, Frisch LM, Gardner TJ, Sarkar S, Hutton JC, Davidson HW. (2009). Novel antigens in type 1 diabetes: the importance of ZnT8. Current diabetes reports. Apr;9(2):105-112.
View at Publisher | View at Google Scholar - Wenzlau JM, Juhl K, Yu L, Moua O, Sarkar SA, et al. (2007). The cation efflux transporter ZnT8 (Slc30A8) is a major autoantigen in human type 1 diabetes. Proceedings of the National Academy of Sciences. Oct 23;104(43):17040-17045.
View at Publisher | View at Google Scholar - Williams CL, Long AE. (2019). What has zinc transporter 8 autoimmunity taught us about type 1 diabetes?. Diabetologia. Nov;62(11):1969-1976.
View at Publisher | View at Google Scholar - Yang L, Luo S, Huang G, Peng J, Li X, Yan X, et.al. (2010). The diagnostic value of zinc transporter 8 autoantibody (ZnT8A) for type 1 diabetes in Chinese. Diabetes/metabolism research and reviews. Oct;26(7):579-584.
View at Publisher | View at Google Scholar

 Clinic
Clinic
