Research Article | DOI: https://doi.org/10.31579/2835-835X/125
Saudi Wild Rumex Vesicarius L Discarding Biofilm Pathogens Interrupting Wound Healing
*Corresponding Author: Sherifa Mostafa M. Sabra S. Const., Prof., Dr. Microbiology.
Citation: Sherifa Mostafa M. Sabra, (2026), Saudi Wild Rumex vesicarius L Discarding Biofilm Pathogens Interrupting Wound Healing, Clinical Trials and Case Studies, 5(2); DOI:10.31579/2835-835X/125
Copyright: © 2026, Sherifa Mostafa M. Sabra. This is an open-access artic le distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 27 March 2026 | Accepted: 03 April 2026 | Published: 24 April 2026
Keywords: Rumex vesicarius L. Staphylococcus aureus, Klebsiella pneumonia, Bacillus subtilis, Pseudomonas aeruginosa, Staphylococcus epidermidis, Proteus vulgaris, Enterococcus faecalis, Actinobacter baumannii, Serratia sp., and Candida albicans.
Abstract
Rumex vesicarius is a Saudi wild plant growing on the mountain, named "Hummayed" or "Hammad". The chemical constituents were anthra-quinones and flavo-noids. It was in traditional medicine list as wound treatment. The goal was to practical study the benefits of Saudi wild Rumex vesicarius extract and its effect on biofilm pathogens. The methodology was for testing Saudi wild Rumex vesicarius extract as antibiofilm. The results first group was included biofilm pathogens Staphylococcus aureus, Klebsiella pneumonia, and Bacillus subtilis. They were highly discarding, the average at day 1 were (0.131, 0.136, and 0.135), at day 2 were (0.090, 0.089, and 0.093), and at day 3 were (0.006, 0.005, and 0.003). The second group was moderately affected were included Pseudomonas aeruginosa, Staphylococcus epidermidis, and Proteus vulgaris. The average at day 1 were (0.279, 0.276, and 0.277), at day 2 were (0.180, 0.183, and 0.187), and at day 3 were (0.071, 0.076, and 0.072). The third group were included Enterococcus faecalis, Actinobacter baumannii, Serratia sp., and Candida albicans. They were less affected, at day 1 were (0.321, 0.326, 0.322, and 0.330), at day 2 were (0.242, 0.245, 0.247, and 0.250), and at day 3 were (0.121, 0.126, 0.128, and 0.130). The conclusions were Saudi wild Rumex vesicarius extract capable discarding biofilm pathogens and biofilm and accelerating wound healing. That recommended use of Saudi wild Rumex vesicarius medicinal plant adhering to pharmaceutical requirements in wound healing and treatment.
Literature Review:
Rumex vesicarius L is a Saudi wild plant growing on the mountain, named "Hummayed" or "Hammad", is from Polygonaceae family. The chief chemical constituents were anthra-quinones and flavo-noids. It was in traditional medicine list as wound treatment [1]. Rumex vesicarius bioactive compounds were phenolic mixes: 8-C-gluco-syl-apigenin, 8-C-gluco-syl-luteolin, 6-C-hexo-syl-quercetin, 3-O-rutino-syl-quercetin, 7-O-rhamno-hexo-syl-diosmetin, 7-O-rhamno-acetyl-hexosyl-diosmetin, catechin, epicate chin, ferulo-hexoside, 6-C-gluco-syl-naringenin, epicatechin gallate, 6-C-gluco-syl-catechin, and epi-gallocatechin gallate. 6-C-gluco-syl-naringenin was the major compound [2]. Rumex vesicarius contained phyto-components; totally, 211 residents, the uppermost 61 phyto-constituents, had p-coumaric acid, ferulic acid, chromone, catechin, and emodin [3]. Rumex vesicarius had 44 chemical components were 99.99%. The major were fatty acids and lipids (51.36%), oxyge-nated hydro-carbons (33.59%), amines (7.35%), carbohydrates (6.06%), steroids (1.21%), and alkaloids (0.42%). The major components were 1,3-di-hydroxy-propan-2-yl oleate (oxy-genated hydro-carbons, 18.96%), ethyl 2-hydroxy-cyclo-hexane-1-carboxylate (ester of fatty acid, 17.56%), and 2-propyl-tetra-hydro-2h-pyran-3-ol (oxygenated hydro-carbons, 11.18%) [4]. Rumex vesicarius had medicinal importance of its chemical composition, contains many bioactive substances, poly-phenols, total phenolic, flavo-noids, and condensed tannins, had anti-bacterial properties [4].
Microbial biofilms recognized on living and nonliving surfaces was not easily eradicated, biofilm cells are more resistance to antimicrobial and delay wound healing [5]. Medicinal plants extract had antibiotic, antibiofilm, and antimicrobial properties against biofilm bacteria concerning eradication of resistant biofilm [6]. Biofilms are linked to 80% of microbial infections, and was widely acknowledged the growth of microorganisms within biofilms can significantly bolster their resistance to antimicrobial agents. Antimicrobial therapies frequently prove ineffective in biofilms eradicating from infection sites [7].
Rumex vesicarius extract showed antibacterial and antibiofilm action [8]. Rumex sp. extracts were antibacterial agents against gram‐positive and gram‐negative bacteria [9]. Rumex vesicarius extract had broad spectrum antibacterial activity against tested organisms. The minimal inhibitory concentration was 62.5–125 mg/mL for gram positive, for gram negative was 125–250 mg/mL. The bactericidal concentration was 250–500 mg/mL for gram-positive and was 500–1,000 mg/mL for gram-negative [9].
Rumex vesicarius extract assessed antibiofilm activity over Pseudomonas aeruginosa biofilm. The minimum biofilm inhibitory concentration (MBIC) was 250 mg/ml. The extract contained antibiofilm Bicyclo (3.1.1) heptan- 3 -one, 2, 6, 6- trimethyl, Bicyclo (3.1.1) heptan, 6, 6- di-methyl and Eucalyptol. There were connections between antibiofilm activity and extracts concentration after 48 and 72 hours [10]. Rumex vesicarius extract inhibited Pseudomonas aeruginosa biofilm formation [10]. It had antibacterial inhibitory on Bacillus subtilis MTCC121, Pseudomonas aeruginosa MTCC 741, and had exhibited anti-biofilm activity [9]. Rumex vesicarius extract had action against gram‐positive bacteria Staphylococcus aureus and Bacillus subtilis and gram‐negative bacteria Pseudomonas aeruginosa [9]. It had antibacterial activity against Staphylococcus aureus (17.83 ± 0.67) [9]. had antibacterial effect on Bacillus subtilis (ATCC 10907), Staphylococcus epidermidis (ATCC 12228), Staphylococcus aureus (ATCC 29213) and Pseudomonas aeruginosa (ATCC 27853) [1]. Rumex vesicarius extract had antibiofilm on Pseudomonas aeruginosa, Klebsiella pneumoniae, and Staphylococcus aureus [8]. It had antibiofilm against Staphylococcus epidermidis, Staphylococcus aureus, Proteus vulgaris, Pseudomonas aeruginosa, and Klebsiella pneumoniae [1]. Anti-Actinobacter baumannii activity was confirmed by Rumex sp. extracts at range 1-4 mg/mL. That showed bacteriostatic and bactericidal activity against MDR Actinobacter baumannii were isolated from wounds [11].
The antifungal activity of Rumex vesicarius extract attributed to biologically residents of active compounds [12]. Rumex sp. extract had antifungal possessions of main biologically compounds [13]. Rumex sp. extract had anti microbial activity against fungal pathogens, Candida albicans [14]. Antifungal activity of Rumex sp. extracts possessed activity against Candida sp. [15].
The goal was to practical study the benefits of using Saudi wild Rumex vesicarius extract and its effect on biofilm pathogens to speed up wound healing and to establish the plant as a medicinal plant..
Methodology:
Peel Extract:
Fresh sample of Saudi wild Rumex vesicarius was gathered from the mountain, and it was identified. The sample was dried in shadow for week, then it was beached by (Electric-Sterile-Grinder). 100 g powder was saturated in 1,000 mL 80% methanol for 3 days in shady at room temperature with even mixing. The result crude extract was concentrated by (Rotary-Vacuum-Evaporator) [9].
Biofilm Pathogens:
Biofilm pathogens were collected from "Private-Microbiology-Lab.", which were isolated and identified from "un-healing, and delay healing wounds samples". They were included Staphylococcus aureus, Staphylococcus epidermidis, Bacillus subtilis, Enterococcus faecalis, Klebsiella pneumoniae, Pseudomonas aeruginosa, Proteus vulgaris, Actinobacter baumannii, Serratia sp., and Candida albicans. They were cultured in "Nutrient-Broth" overnight, and then were sub-cultured on "Nutrient-Agar" overnight. They were adjusted for pure cell suspension by "Peptone-Water" were adjusted to "McFarland-Turbidity"; "0.5=106 CFU/mL" [16].
Antibiofilm Method:
Biofilm pathogen cell suspension 0.2 mL + Saudi wild Rumex vesicarius extract 0.2 mL were additional in "Microtiter-Platter", (Falcon, USA). Then was incubated (1, 2, and 3) days at 37 °C. The biofilm in plate was fixed by sodium acetate for 30 min., and was stained by "Crystal-Violet" for 30 min. Then was washed by deionized water and was reserved for drying. The estimation of "Optical densities"; (OD) by "Micro Enzyme Linked Immunosorbent Assay Auto Reader"; (OD, Wavelength 570 nm) [17].
Statistical Analyses
The results were giving triple trials, were existed as mean with standard errors. These final results were intended by "Microsoft-Excel-Software" [18].
Results and Discussions:
Biofilm Pathogen | Time | ||
Day 1 | Day 2 | Day 3 | |
Staphylococcus aureus | 0.131±0.001 | 0.090±0.002 | 0.006±0.002 |
Klebsiella pneumonia | 0.136±0.002 | 0.089±0.003 | 0.005±0.002 |
Bacillus subtilis | 0.135±0.001 | 0.093±0.002 | 0.003±0.002 |
|
|
|
|
Pseudomonas aeruginosa | 0.279±0.002 | 0.180±0.001 | 0.071±0.002 |
Staphylococcus epidermidis | 0.276±0.003 | 0.183±0.002 | 0.076±0.003 |
Proteus vulgaris | 0.277±0.002 | 0.187±0.001 | 0.072±0.002 |
|
|
|
|
Enterococcus faecalis | 0.321±0.001 | 0.242±0.002 | 0.121±0.001 |
Actinobacter baumannii | 0.326±0.002 | 0.245±0.002 | 0.126±0.002 |
Serratia sp. | 0.322±0.001 | 0.247±0.001 | 0.128±0.001 |
Candida albicans | 0.330±0.001 | 0.250±0.001 | 0.130±0.001 |
Table 1: Estimation of anti-biofilms formation after exposed to Saudi wild Rumex vesicarius extract
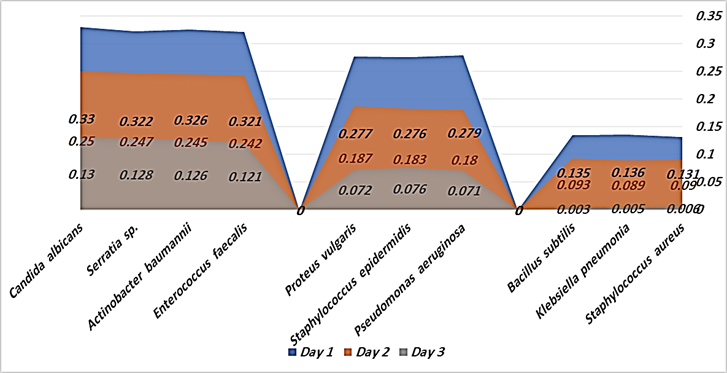
Figure 1: Estimation of anti-biofilms formation after exposed to Saudi wild Rumex vesicarius extract
Table (1), and Figure (1) were showed estimation of anti-biofilms formation after exposed to Saudi wild Rumex vesicarius extract. The biofilm pathogens were divided into three groups based on the results of discarding biofilm pathogens. The first group was included biofilm pathogens Staphylococcus aureus, Klebsiella pneumonia, and Bacillus subtilis. They were highly discarding affecting by Saudi wild Rumex vesicarius extract. The average "Optical Density" results which indicated the biofilm present. That were being within three days, the results at day 1 were (0.131, 0.136, and 0.135), at day 2 were (0.090, 0.089, and 0.093), and at day 3 were (0.006, 0.005, and 0.003). Biofilm pathogens and biofilms were almost completely discarded [1, 8-9, 11]. This was indicated the presence of active ingredients in the Saudi wild Rumex vesicarius extract capable of discarding biofilm pathogens and the biofilms itself [1-9].
The second group was moderately affected by Saudi wild Rumex vesicarius extract, within three days, the biofilm pathogens were not completely discarded.
The second group was included biofilm pathogens Pseudomonas aeruginosa, Staphylococcus epidermidis, and Proteus vulgaris. They were moderately discarded affecting by Saudi wild Rumex vesicarius extract. The average "Optical Density" results which indicated the biofilm present. That were being within three days, the results at day 1 were (0.279, 0.276, and 0.277), at day 2 were (0.180, 0.183, and 0.187), and at day 3 were (0.071, 0.076, and 0.072). This was indicated that an additional day is needed to completely discarding biofilm pathogens and biofilms [1,8-10]. These results were explained the strength of the active ingredients in Saudi wild Rumex vesicarius extract [1-9].
The third group were included biofilm pathogens Enterococcus faecalis, Actinobacter baumannii, Serratia sp., and Candida albicans. They were less affected by Saudi wild Rumex vesicarius extract. The average results were at day 1 were (0.321, 0.326, 0.322, and 0.330), at day 2 were (0.242, 0.245, 0.247, and 0.250), and at day 3 were (0.121, 0.126, 0.128, and 0.130). This was indicated that an additional more than one day is needed to complete discarding biofilm pathogens and biofilms [8-9, 11-12,15]. This was indicated that Saudi wild Rumex vesicarius extract components are potent in discarding biofilm pathogens and biofilm, but require a longer period of time [1-9].
Based on the results, these were biofilm pathogens cause delayed wound healing and require a treatment by medicinal plants. The research demonstrated that Saudi wild Rumex vesicarius extract active ingredients had the ability to eliminate biofilm pathogens and biofilms. With the specific type of biofilm pathogens responsible for this effect, were varying depending on the biofilm pathogens type. This will lead to faster wound healing and improved health [1-9].
Conclusions:
The laboratory experiments showed Saudi wild Rumex vesicarius extract contains active ingredients capable of discarding biofilm pathogens and biofilm and accelerating wound healing.
Recommendations:
The use of Saudi wild Rumex vesicarius as a medicinal plant adhering to the pharmaceutical requirements in wound healing and treatment. Saudi wild Rumex vesicarius medicinal plants will be reduce the time and reduces the need for chemotherapy.
Acknowledgments:
The grateful were directed to the researchers collaborated on this research.
References
- Fady, M., Rizwana, H., Alarjani, M., Alghamdi, A., Ibrahim, S., et al. (2023). Evaluation of antibiofilm and cytotoxicity effect of Rumex vesicarius methanol extract. Open Chemistry, 21(1), 20220286.
View at Publisher | View at Google Scholar - Sweilam, H., Abd El Hafeez, S., Mansour, A., & Mekky, H. et al. (2024). Unravelling the phytochemical composition and antioxidant potential of different parts of Rumex vesicarius L.: A RP-HPLC-MS-MS/MS, chemometrics, and molecular Docking-Based comparative study. Plants, 13(13), 1815.
View at Publisher | View at Google Scholar - AkASHA, R., HUSSEIN, W., MONI, S., ALAFNAN, A., ALSAIKHAN, A., et al. (2025). Antibacterial Potential of Rumex vesicarius: A Phytochemical Perspective. Oriental Journal of Chemistry, 41(5).
View at Publisher | View at Google Scholar - Salama, A., Al-Faifi, E., Masood, F., & El-Amier, A. et al. (2022). Investigation and biological assessment of Rumex vesicarius L. extract: Characterization of the chemical components and antioxidant, antimicrobial, cytotoxic, and anti-dengue vector activity. Molecules, 27(10), 3177.
View at Publisher | View at Google Scholar - Cavallo, I., Sivori, F., Mastrofrancesco, A., Abril, E., Pontone, M., et al. (2024). Bacterial biofilm in chronic wounds and possible therapeutic approaches. Biology, 13(2), 109.
View at Publisher | View at Google Scholar - Mulat, M., Banicod, S., Tabassum, N., Javaid, A., Karthikeyan, A., et al. (2025). Multiple strategies for the application of medicinal plant-derived bioactive compounds in controlling microbial biofilm and virulence properties. Antibiotics, 14(6), 555.
View at Publisher | View at Google Scholar - Singh, B., Dahiya, M., Kumar, V., Ayyagari, A., Chaudhari, N., et al. (2025). Biofilm and antimicrobial resistance: mechanisms, implications, and emerging solutions. Microbiology Research, 16(8), 183.
View at Publisher | View at Google Scholar - Bouafia, A., Laib, I., Laouini, E., Azzi, M., Alharthi, F., et al. (2025). Comprehensive Bioactivities and Phytochemical Profiling of Rumex vesicarius: Antioxidant Potential, Anti‐Diabetic Properties, and Anti‐Biofilm Effects Under Thermal Treatment. Chemistry & Biodiversity, 22(9), e202500329.
View at Publisher | View at Google Scholar - Sulieman, E., Abdallah, M., Alanazi, A., Idriss, H., Adnan, M., et al. (2024). Bioactive profiling of Rumex vesicarius L. from the Hail region of Saudi Arabia: A study on its phytochemical and elemental analysis, antibiofilm, antibacterial, antioxidant properties, and molecular docking analysis. Frontiers in microbiology, 15, 1421143.
View at Publisher | View at Google Scholar - Oalđe Pavlović, M., Kolarević, S., Đorđević Aleksić, J., & Vuković-Gačić, B. et al. (2024). Exploring the antibacterial potential of Lamiaceae plant extracts: inhibition of bacterial growth, adhesion, invasion, and biofilm formation and degradation in Pseudomonas aeruginosa PAO1. Plants, 13(12), 1616.
View at Publisher | View at Google Scholar - Andrzejczuk, S., Sozoniuk, M., & Sugier, D. et al. (2025). Preliminary Assessment of Arnica montana L. Extract: Antimicrobial Activity Against Acinetobacter baumannii and Biofilm-Related Gene Expression Profiling. Genes, 16(12), 1473.
View at Publisher | View at Google Scholar - Hafaz, F., Soliman, M., Abbas, A., Gebreil, S., & El-Amier, A. et al. (2022). Potential Assessment of Rumex spp. as a source of bioactive compounds and biological activity. Biointerface Research in Applied Chemistry, 12(2), 1824-1834.
View at Publisher | View at Google Scholar - Mohamed Gohar, M., Ezzat, M., Yeskaliyeva, B., Elhawary, S., Kirollos et al. (2025). Rumex Species: Phytochemistry, Pharmacology and Nutritional Potential for Food and Health Applications. Food Science & Nutrition, 13(12), e71300.
View at Publisher | View at Google Scholar - Xiao, D., Sun, H., Li, X., Meng, F., Sun, T., et al. (2024). Rumex japonicus Houtt. leaves: The chemical composition and anti-fungal activity. Journal of Medical Mycology, 34(4), 101513.
View at Publisher | View at Google Scholar - Al-Garadi, A., Qaid, M., Alqhtani, H., Pokoo-Aikins, A., & Al-Mufarrej, I. et al. (2022). In vitro phytochemical analysis and antibacterial and antifungal efficacy assessment of ethanolic and aqueous extracts of Rumex nervosus leaves against selected bacteria and fungi. Veterinary World, 15(11), 2725.
View at Publisher | View at Google Scholar - Yucens, M., Sari, T., Aydemir, N., Öztürk, S., Akman, et al. (2025). Evaluation of pathogens isolated from wound infections and their antibiotic susceptibility profiles in a wound care unit. The Journal of Infection in Developing Countries, 19(09), 1351-1358.
View at Publisher | View at Google Scholar - Monyela, S., Kayoka, N., Olawuwo, S., Ngezimana, W., & Nemadodzi, et al. Anti-quorum Sensing and Cytotoxic Activities of Leaf Crude Extracts of Cannabis' Gorilla Glue 1'. Frontiers in Veterinary Science, 13, 1750799.
View at Publisher | View at Google Scholar - Kiviaho, K., Anttonen, M., Alfthan, H., & Itkonen, O.et al. (2025). Reliable quantification of fecal elastase-1: A study on sample stability, IDK ELISA and IDK Extract® device. Clinica Chimica Acta, 120541.
View at Publisher | View at Google Scholar

 Clinic
Clinic
