Research Article | DOI: https://doi.org/10.31579/2834-5134/023
Recent Advances in The Diagnosis of Well-Differentiated Colon Neuroendocrine Tumors
1 Carol Davila University of Medicine and Pharmacy Bucharest
2 Ion Cantacuzino Clinical Hospital Bucharest, Department of Internal Medicine
3 Elias Emergency University Hospital Bucharest, Department of Oncology
*Corresponding Author: Marilena Stoian, Carol Davila University of Medicine and Pharmacy Bucharest, Romania.
Citation: Marilena Stoian, Gabriel Scarlat, Iuliana Ion M.D. (2023). Recent Advances in The Diagnosis of Well-Differentiated Colon Neuroendocrine Tumors. Journal of Clinical Anatomy, 2(3) DOI:10.31579/2834-5134/023
Copyright: © 2023 Marilena Stoian, This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 29 May 2023 | Accepted: 08 June 2023 | Published: 19 June 2023
Keywords: gep-nens; gep-nets; diagnosis; biochemical markers; imaging; histopathology
Abstract
Neuroendocrine tumors (NETs) are rare tumors that appear with secondary symptoms based on the release of vasoactive peptides and hormones into the blood as an answer to a signal from the nervous system. Neuroendocrine neoplasms of the colon are epithelial neoplasms with neuroendocrine differentiation and are classified in step with the foremost recent WHO 2022 classification as well- differentiated neuroendocrine tumors (NETs) and poorly differentiated neuroendocrine carcinoma- mas (NECs. NETs can be divided into serotonin-producing enterochromaffin-cell neuroendocrine tumor and glicentin-PYY- producing L-cell neuroendocrine tumor. Compared to NECs, NETs are often idle lesions occurring as incidental findings, being generally characterized by indolent course and relatively frequent ab- since of specific symptoms, which underscores the significance of establishing a prompt and accurate diagnosis, colonic neuroendocrine tumors generally presenting as a large tumor with local or distant metastasis (generally liver) at the time of diagnosis.
While utmost colon NETs are considered non-aggressive tumors, a few cases may show a more aggressive clinical course. Clinical/pathological characteristics to select cases at high threat of recurrence/metastases are sadly inadequately consolidated. Diagnosis is based on the combination of clinical presentation, biochemical markers, endoscopy, imaging and confirmed by histopathology. Differential diagnostic for NETs can cause various problems: the rare appendiceal tubular and clear cell variants (which may be confused with non-neuroendocrine cancers). This review presents the updates in established diagnostic tests and tests that require further development, investigation, and validation before use [1,7].
Introduction
Neuroendocrine tumors (NETs) appear from neuroendocrine cells of the diffuse endocrine system. These cells have characteristics of both nerve cells that can receive message from the nervous system and endocrine cells that can synthesize and secrete monoamines, peptides, and hormones). Neuroendocrine cells have no axons or nerve terminals. The electrical signals from the nervoussystem can be change into hormonal signals leading to production of hormones, pep- tides and amines. They are considered rare slow-growing tumors with distinct histological, bio- logical, and clinical characteristics that have increased in incidence and prevalence withinthe last few decades. Colon carcinoid tumors (C-NETs) are well differentiated neuroendocrine tumors of the colon, classified as G1 slow grade, G2-intermmediate grade and G3-high grade (WHO classification) [1].
Because NENs often present as small lesions and in variable anatomical localization, the diagnostic of NENs can be challenging. The gold standard remains histopathology, which should contain neuro- endocrine specific markers, such as chromogranin A and also an estimate of the proliferation by Ki-67 which is very important for treatment selection and prognostication. Lately, the measurement of multiple NEN-related transcripts has been introduced, will play a key role, and seems to be superior to serum markers such as chromogranin A. The diagnostic workup of NENs was therefore revolutionized by the introduction of nuclear medical procedures able to detect the presence of somatostatin receptors (SSTRs), which are typically overexpressed in well- differentiated NEN cells. According to studies from recent years, SSTR PET/CT imaging is superior to SSTR scintigraphy or conventional anatomic imaging (US, dCT or MRI) for the assessment and diagnosis of well-differentiated NETs. SSTR PET/CT presents the advantage that it can detect the primary tumor site, it can frequently demonstrate additional lesions not captured by conventional imaging, performing in more accurate evaluation of disease extension. All these characteristics lead to relevant changes in management in about one-third of patients [5,8].
In fact, diagnosis is usually determined either at biopsy of mass or after surgical resection and most patients show advanced stage at diagnosis [1].
The purpose of this review is to present the novel diagnostic modalities of well-differentiated colon NETs that have been raised over the last years.
Diagnosis
Because the prevalence of well-differentiated neuroendocrine tumors of colon has in- creased over the last decades, new methods of diagnosis and the improvement of existing ones have been sought, creating new methods of investigation and the imposition of new appropriate and much more effective diagnostic algorithms. Table 2 summarizes current diagnostic tools for colon NETs and fig.2 shows a diagnostic algorithm.
Clinical presentation
GI tract NETs are usually asymptomatic, they do not manifest specific symptoms initially but only display later symptoms that are related to tumor growth or metastases, with even complete colon obstruction and episodes of diffuse abdominal colicky pain and altered bowel habits (incomplete) an acute episode of ileus (complete). The colonic locations often present with large lesions (more than 2 cm in diameter) with multiple metastases and have poor differentiation (grade 3) and aggressiveness. It manifests as adenocarcinoma, while carcinoid syndrome is extremely rare [7].
Most colonic NETs (70%) are localized in the right colon, particularly in the cecum. Be- cause the right colon has an increased diameter than the left one, the patients present no symptoms until the NET size becomes large, the average size of the NETs being about 5 cm and most having local or distant metastasis at the time of diagnosis. Patients usually have the following symptoms and signs: abdominal pain due to mass effect or tumor-induced desmoplastic reaction, gastrointestinal bleeding, and weight loss. (3)
Chromogranin A concentration
Chromogranins (designated as chromogranin A [CgA], chromogranin B [CgB], and chro- mogranin C [CgC]) are proteins that are stored and released with peptides and amines in a variety of neuroendocrine tissues. High blood concentrations of chromogranins are associated with well- differentiated NETs, including carcinoids, and it has been found that they increase the larger the tumor becomes [2].
Serum CgA has been the general biomarker for well- differentiated NETs for a long time, but its diagnostic accuracy is now under debate, consensus-based guidelines having decreasingly played down their part in clinical care for the following reasons:
False positive results occur in some circumstances including drugs (they are especially common in patients who are taking proton pump inhibitors), inflammatory bowel disease, heart and renal failure, malignancy [7], uncontrolled hypertension, pregnancy, hypergastrinemia, chronic atrophic gastritis, steroid treatment, or glucocorticoid excess [8].
Levels of CgA secretion can easily vary daily in healthy subjects and those with NETs, resulting that CgA is a relatively nonspecific marker for NETs [2].
There is no international standard for CgA assays, multiple CgA tests use different assays, have widely divergent normal thresholds and different degrees of accuracy [2].
Tumor cell type and histologic differentiation can influence the levels of CgA, studies showing that these levels are elevated in patients with NETs of midgut origin [6].
Recent studies show that CgA levels significantly elevated levels are are most likely met only in NENs, with the exception maybe of patients receiving PPIs. Regarding this aspect, patients should interrupt administration of PPIs, if this is possible and safe for the patient and leaving a clearance of at least 3 half- lives, prior to testing [8,28].
In order to combat the fact that there is no recognized international standard for CgA assay and variations in assay types may impact results, it is advisable to perform serial measurements using the same assay and reference laboratories should be preferred [8].
NETest The NETest index is a mRNA genomic biomarker in peripheral blood, comprised of 51 marker genes, which uses PCR technology and multianalyte algorithmic analyses to test for neuroendocrine neoplasms, to differentiate tumors from controls, to differentiate stable disease from progressive disease and to accurately predict the development of NET metastases. The most important advantages of the NETest are:
- it can identify all types of NETs,
- it can detect early-stage nonmetastatic disease, showing the percentage of positivity of the genes involved. The results show a scale from 0% to 100% as an activity index. The cutoff point is 20%. An index between 20% and 40% indicates stable disease, whilean index above 40% indicates progressive disease,
- NETest is significantly more accurate than CgA assays in differentiating NETs from con-trols and in detecting disease progression,
- its diagnostic accuracyis high (99%) compared to thatof CgA (21%-36%),
- it can diagnoserecurrence and may determine prognosis.
All these advantages make the NETest index the most successful multianalyte biomarker as passed to date in the management of NENs [6,7,8,11,12,19,28- 34].
Genetic and molecular testing
Patients with multiple endocrine neoplasia’s such as hyperparathyroidism and/or pituitary tumors, a family history of NENs or associated diseases and featuressuspicious of a hereditary disease and young patients (<40>
Molecular markersanalysis is now essential for establishing an accurate diagnosis. Some of these analyses require investigation of somatic (acquired) genetic alterations, gene, or protein expression, oreven circulating tumor markers. Entity-defining for well-differentiated NETs is the mutations in MEN1, DAXX and ATRX [6]. It is very important that well-differentiated NETs have intact p53 and demonstrate no Rb loss in immunohistochemistry [6,19].
The molecular pathways driving midgut NETs are not very well understood yet as mentionedin recent studies. However, mutations of chromosome 11q, affecting the tumor suppressor gene succinate dehydrogenase complex subunit D (SDHD) have been associated with midgut NETs, also mutations in cyclin dependentkinase inhibitors (CDKNs) have an association with midgut-derived NETs, with focal deletion peaks at 9p21 (CDKN2A) and 12p13 (CDKN1B). It has been found that the number of alterations were significantly higher in metastases comparedto primary tumors,suggesting an accumulation of acquired genetic changes as tumors progressed. Losses in 18q and 11q have been found in primaryand metastatic tumors.In metastatic diseasewere detected losses in 16q and 4 [6,19,23,24].
It is believed that hindgut NETs develop via different molecular pathways comparedto midgut NETs. There was a significant correlation between Ki-67 levels and glucose transporter 2 (GLUT2) scoresand O₆-methylguanine DNA methyltransferase (MGMT) scores in hindgutNETs [6,19,23,24].
Histopathology and Immunohistochemistry (IHC)
Histological diagnosis is obligatory in all cases and can be carried out on resectionsamples or core biopsies in advanced disease.European Neuroendocrine Tumor Society (ENETS)recommends histopathological confirmation as the gold standard for the diagnosisof NENs. Histo morphological growth pattern and cytology highlighted on the hematoxylin eosin (HE)-stained tissue can put the diagnosis of NENs. Table 1 shows the WHO 2022 classification system for neuroendocrine neoplasms (NEN) based on the histopathological findings.The neuroendocrine phenotype is proven by the immunohistochemical discovery of the neuroendocrine markerssyn- aptophysin and/or chromogranin A (CgA). Because of their low grade of specificity, neuron-specific enolase (NSE)and CD56 markersare not recommended even if they are frequently positive in GEP-NENs [4,8,19,24,27-31].
In routinelyprocessed tissue sections,these neuroendocrine cells can be identified histochemically by their argentaffin or argyrophil properties or immunohistochemistry staining for ge neric neuroendocrine markers as chromogranins, synaptophysin, neuron-specific enolase (NSE) and PGP9.5 that relate to their neurosecretory granules, cytosol, or vesicles. Specificcell types like serotonin- producing EC cells are best identified by the immunohistochemical localization of their secretory products in their cytoplasm [10].
C-NETs (35% of colonicNENs) arise from Kulchitsky cellsor enterochromaffin cells located within the crypts of Lieburkuhn of colon and typically show enterochromaffin (EC)-cellfeatures (insular architecture, serotonin production and CDX2 positivity). Colon NETs are comprised of a bland appearing monotonous population of cells with roundnuclei and finely stippled salt-and-pepper chromatin. G1 or G2 NETs are usually immunoreactive for neuroendocrine mark ers (synaptophysin, chromogranin). In a minority of cases EC-cell NETs are immunoreactive for neuroendocrine markers,serotonin, SSTR2A and CDX2 and L-cell NETs are immunoreactive for neuroendocrine markers (synaptophysin>chromogranin) and PYY, glicentin, GLP-1, GLP-2, SSTR2A and PAP. At diagnosis, about 30-40% are metastatic to the liver, nodes, mesentery, and peritoneum. Indeed, C-NETs have one of the lowest median survival rates of all NETs (5-year survival rates according to stage are: 80% for stage I or II; 50% for stage III, 10% for stage IV). C- NETs are usually easily identified in histology [1,3,9].
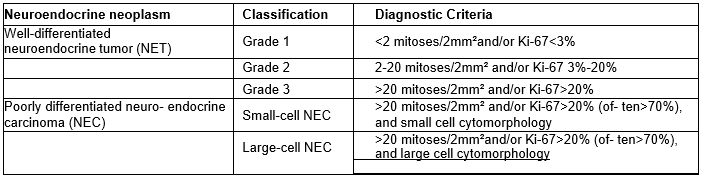
Table 1: WHO 2022 classification system for neuroendocrine neoplasms (NEN)
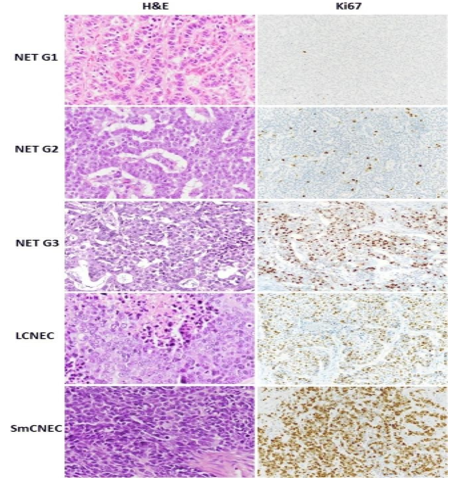
Figure 1: Morphology and Ki67 proliferation index of differentNEN types of the digestive system.
Immunohistochemistry (IHC) should be performed in addition to the conventional histopathological analysis, to assess the tumor phenotype and Ki-67 (MIB1) which is mandatoryto grade the C-NETs fromG1 to G3 according to the WHO classification. (Fig.2)In assessing the differentiation of NENs, is critical to evaluate Ki-67 cell proliferation associated nuclear marker. The Ki- 67 index based on assessment of 2000 cells and the number of mitotic figures per 2mm² should be reported. To determine the Ki-67 proliferation index, at least 500 cells in the regions of highest labelling, known as “hotspots”, are counted. These areas are identified via scanning magnification. When areas with two varyingproliferation indices are present in a particular sample, the area with higher proliferation index is selected for grading purposes[4,8,10].
Imaging Computed tomography (CT)
Computed tomography represents the basic radiological method for NET imaging because of its large availability, standardized reproducible techniqueand generally high level of diagnostics. CT has the advantage of being the best modalityto assess vascular infiltration and to be very helpful in the pre-operative setting. Unfortunately, CT investigation has some limitations such as:
- small metastatic lymph nodes (<1>
- sensitivity for bone metastases is poor ranging from 46% to 80%, with a median value at61%.
- small peritoneal metastases may be difficult to visualize.
CT scanning has a specificity to detect NETs between 61%–93% and a specificity between 71%–100%. Liver metastases (LMs) are detected at an average rate of 79% (73%–94%). For extra- abdominal soft tissue metastases, the sensitivity is between 60%– 100% with an averageof 70% and a specificity range between 87%–100%,with an average of 96% [4,8,19].
Magnetic resonance imaging(MRI)
This imaginginvestigation is advantageous for examination, it has the advantage of no exposureto radiation, higher tissue resolution than CT and is usuallypreferred in the initial staging,for the preoperative imaging work-upand for assessing bone metastases. MRI may be preferred as the imaging modality of choice or complementary to CT and for patients who are allergic to iodine contrast agents.
Currently, diffusion-weighted imaging (DWI) with MRI (DW-MRI), based on their restricted movement of water in highly cellular tissues such as in tumor’s, is routinely applieddue to its advantages:
- It facilitates lesion detection.
- It allows detectionof subtle neoplastic tissue changes.
It is highly sensitive in detecting livermetastases.
The MRI sensitivity to detect NETs has an average of 79% (54%–100%), with almost similardetection rates of 76% (61%–95%). For liver metastases, the sensitivity has an average of 75% (range 70%–80%) with a maximum specificity of 98%. The mean sensitivity of MRI for detection of liver metastases is 91% (range 82%–98%) as compared with CT with a mean sensitivity of 83% (range 75%–98%). Regarding the imaging of the bones and the brain, MRI is also superior to CT and the accuracy of DW- MRI for the detection of primary NETs and metastatic disease is compa rable to PET/CT. MRI is also superior to CT when using the hepatocyte specific contrast media for characterizing liver lesions [4,8].
SSTR scintigraphy (SRS)
This kind of investigation should be carried out only when PET-CT is not
available but is considerably less sensitive. SRS must include cross-sectional imaging by single photon emission CT (SPECT) combined with CT (SPECT-CT). The power of PET-CT has a higher detection rate of lymph node, bone, peritoneal lesions, and unknown primary tumors. (4,19,22)
Colonoscopy
Colonoscopy represents a good diagnostic investigation for colonic NETs (method to diagnose small NETs with 86% (range 82%–93%) sensitivity and 92% (range 86%–95% specificity), allowing biopsy using fine needle aspiration for cytology or a cutting needle for histopathological diagnosis and resection of the primary NET, when indicated. Unfortunately, colonoscopy is an invasive procedure associated with adverse effects and misleading biopsies (NETs are sub- epithelial lesions) [4,8,24,26,27,29].
Nuclear medicine
Somatostatin receptor (SSTR) positron emission tomography (PET) in combination with CT (PET-CT)
Somatostatin is a cyclic peptide which has strong regulatory effects in the body, its action being mediated through membrane-bound receptors which are expressed in high volumes in neuroendocrine cells and NENs. sst1-sst5 are five subclasses that have been cloned [5].
SSTR PET/CT has demonstrated several advantages over scintigraphy imaging (higher spatial resolution, more favorable biodistribution at liver/bowel level, semi-quantitative analysis) and is currently recommended by all guidelines [5,15].
Imaging by ⁶⁸Ga/⁶⁴Cu-DOTA-somatostatin analogue (SSA) positron emission tomography (PET) in combination with CT (PET-CT) provides high sensitivity for imaging of most types of NET lesions and should be part of the tumor staging, preoperative imaging, and restaging [4,19,25].
The radiopharmaceuticals used for SSTR PET/CT present a common structure: a positron- emittingisotope (⁶⁸Gallium, ⁶⁸Ga), a chelate (DOTA)and the SSTR ligand (NOC, TOC, TATE) which corresponds to three clinically employed tracers showing variable affinity for the SSTR.
subtypes: [⁶⁸Ga] Ga—DOTA-TOC, [⁶⁸Ga] Ga— DOTA-NOC and [⁶⁸Ga] Ga—DOTA-TATE. ⁶⁸Ga is a positron-emitting radionuclide that can only be applied for diagnostic imaging, its short half- life (68 min) making it unsuitable for therapeutic purposes[5]
⁶⁸Ga -DOTATATE PET/CT con- firms the diagnosis of NET, localizes unknown primary NETs and identifies additional sites of disease that can divide surgical versus nonsurgical patients [7,25] A study from 2022emphasizes the fact that on the diagnostic side, [⁶⁸Ga]Ga-DOTA peptides PET/CT (SSTR PET/CT) represents the gold standardfor imaging well differentiated SSTR-expressing neuroendocrine tumors (NETs) [5,13,14,16].
For diagnostic purposes, DOTATATE was also labelled with ⁶⁴Cu, ⁶⁴Cu-DOTA offering a longer radionuclide half-life(12.7h of ⁶⁴Cu vs 68 min of ⁶⁸Ga), shorter positronrange in tissue(mean 0.6 vs 3.5 mm) and lower positron branching ratio (0.17 vs 0.89),resulting in higher TBR (tumor-to-background ratio) at delayed images [5].
[¹⁸F] FDG PET/CT is useful in higher grade NENs (NET G2 with Ki-67>10% and NET G3) for more accuratedisease characterization and prognostication. Promisingemerging radio- pharmaceuticals include somatostatin analogueslabelled with ¹⁸F (to overcome thelimits imposedby
⁶⁸Ga) and SSTR antagonists (for both diagnosis and therapy). The ESMO 2020 guidelines rec commends the use of double-tracer ([¹⁸F] FDG+SSTR) PET/CT for all G2-G3 NETs in order to achieve the optimaldiagnostic [5,15,16,18,19].
According to the European Association of Nuclear Medicine (EANM) guidelines (last up- dated in 2017),SSTR PET/CT is recommended as the first choice to study patients with foregut and midgut NETs and it is generally considered to be the second choice for hindgut NETs (secondto [¹⁸F] FDG) [5].
The sensitivity to detect NET disease by 68Ga- DOTA-SSA-PET-CT ranges between 64%– 100% with an average of 92% and a specificity average of 95% (range 83%–100%). It has a sensitivity to detect bone metastases between97%–100% and a specificity between 92%–100%. The use of PET with fluoro-deoxy-glucose (FDG) is optionalin NENs. FDG has higher glucose metabolism and less SSTR expression than the low-gradeNETs and for this reasonthe tracer of choice for G3 and high G2 NETs is FDG. SSTR imaging plus FDG-PET-CT has been shown to be complementary for lesion detection. A worse prognosis for NETs is given when findings at PET- CT are FDG positive. Submitting all NET G2/G3patients to PET-CT with FDG and 68Ga- DOTA-SSA (DOTATOC/DOTATATE/DOTANOC) it can obtain the best diagnostic and prognostic information. (4,13- 18,21,26,27) Regardingthe diagnosis of NETs, severalguidelines recommend SSTR PET/CT for [5,30,31].
- Detection of primary occult site in patients with demonstrated neuroendocrine metastasis or with increased specific tumor markers, with negative conventional imagingfor primary lesions.
- Staging after the histological diagnosis of NET and prior to plannedsurgery.
- Localization of a primarytumor (known metastatic disease but an unknown primary)
- Detectionof a suggestive NET mass not submittedfor endoscopic or percutaneous biopsy.
- Evaluation of patients with biochemical evidence and symptoms of a NET but without histological diagnosis
- Radiolabeled peptide 99mTcEDDA/HYNIC-TOC can represent anothergood alternative for diagnosing NETs in countries and clinics that do not have PET equipment and 68Ga generators, 99Tc being easily maneuvered and radiation for patients being relatively small. The study’sresults show a high sensitivity (90.5%), even if lower than 68Ga-DOTA, but with good specificity, ac- curacy, positive and negative predictive values, widely availableand cheaper [20].
| Modality | Indication | Strengths | Limitations |
| Histopathology | -must be done in all colon NETs, when possible | -goldstandard diagnosis procedure | -needs an expert pathologist |
Chromogranin A serum |
-usedas diagnosis and follow-up |
-very good, studied biomarker -canbe used in all colonNETs | -it has a moderately sensitive and variable specificity -false positive results because of several factors |
Computed tomography (CT) |
-used for diagnosis, staging, follow-up, and assessment of treatment response |
-widely available -very goodestablished modality. -best modality to detect vascular infiltration -useful in the pre-operative setting | -lack of international standard assay -radiation exposure -variable sensitivity -less accurate in the diagnostic of colonic NETs but still important for staging |
Magnetic resonance imaging (MRI) | -similar to CT -modality of choice or complementary to CT | -not contraindicated in patient allergic to iodinecontrast -no radiation expo-sure | -less available than CT -contraindicated in patients with metallic implants |
⁶⁸Ga-DOTA peptides |
-investigation of choice for well -differentiated NETs | -mean sensitivity and specificity: 88%- 93% and 88%-95% respectively -sensitive in detecting even subtle lymph node or small peritoneal metastases -unaffected using somatostatin analogs before ex- amination -lower exposure than classical scintigram- |
-still not broadly available -more expensive than other modalities |
| ¹⁸FDG PET/CT | -usefulin higher | -accurate diagnosis | -not broadly available |
| gradeNETs (G2 | |||
| and G3) and in | -more expensive | ||
| rapidlyprogressive | than other modalities | ||
| cases | -falsely positive | ||
| results in active in | |||
| inflammation or infection | |||
| 99mTcEDDA/HYNIC- TOC |
-investigation of | -widely available |
-lowersensitivity |
| choice | -cheaper than 68Ga- | compared with gold | |
| DOTA-TATE | standard 68Ga- | ||
| -good sensitivity | DOTA-TATE | ||
| (90.5%), specificity | |||
| and accuracy | |||
| Colonoscopy | -investigation of | -allowsbiopsy | -invasive procedure |
| Choice forcolonic | -primary NET can be | -associated withad- | |
| NETs | resected when indicated | verseeffects | |
| -biopsies may be | |||
| misleading as NETs | |||
| are subepithelial lesions |
Table 2: Current diagnostictools in colon NETs
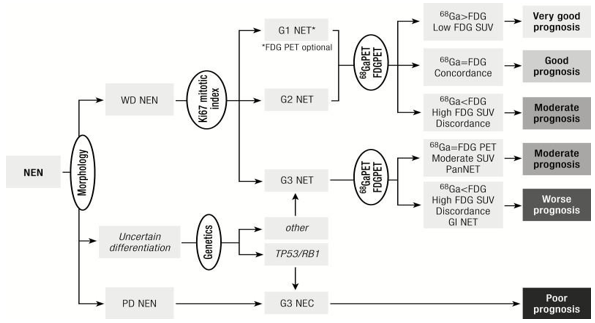
Figure 2: Diagnostic algorithm
Conclusions
Colon NETs are complex tumors which require a vast, accurate and early diagnostic approach to influence prognosis and patients’ treatment. Histopathology is and will probably remainthe gold standarddiagnostic in the future. CT and MRI are also improving the diagnostic, regarding their advances. The revolution in the field is added by the advancesin nuclear medicine, ⁶⁸Ga- DOTA PET/CT giving an unprecedented diagnostic accuracy, unfortunately still being limitedby its high-cost and availability. Multianalyte biomarkers, like NETest which is standardized and not influenced by age, gender, or medication, are promising diagnostic tools which also provide accurate information about completeness of surgical resection, the presence of residual disease and predict the therapeutic efficacy.
References
- Volante, M., Grillo, F., Massa, F., Maletta, F., Mastracci, L., Campora, M., ... & Papotti, M. (2021). Neuroendocrine neoplasms of the appendix, colon and rectum. Pathologica, 113(1), 19.
View at Publisher | View at Google Scholar - Di Giacinto, P., Rota, F., Rizza, L., Campana, D., Isidori, A., Lania, A., ... & Baldelli, R. (2018). Chromogranin A: from laboratory to clinical aspects of patients with neuroendocrine tumors. International Journal of Endocrinology, 2018.
View at Publisher | View at Google Scholar - Ahmed, M. (2020). Gastrointestinal neuroendocrine tumors in 2020. World journal of gastrointestinal oncology, 12(8), 791.
View at Publisher | View at Google Scholar - Pavel, M., Öberg, K., Falconi, M., Krenning, E. P., Sundin, A., Perren, A., & Berruti, A. (2020). Gastroenteropancreatic neuroendocrine neoplasms: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Oncology, 31(7), 844-860.
View at Publisher | View at Google Scholar - Ambrosini, V., Zanoni, L., Filice, A., Lamberti, G., Argalia, G., Fortunati, E., ... & Fanti, S. (2022). Radiolabeled somatostatin analogues for diagnosis and treatment of neuroendocrine tumors. Cancers, 14(4), 1055.
View at Publisher | View at Google Scholar - Alexander, E. S., & Ziv, E. (2023). Neuroendocrine Tumors: Genomics and Molecular Biomarkers with a Focus on Metastatic Disease. Cancers, 15(8), 2249.
View at Publisher | View at Google Scholar - Pavlidis, E. T., & Pavlidis, T. E. (2022). Molecular factors, diagnosis and management of gastrointestinal tract neuroendocrine tumors: An update. World Journal of Clinical Cases, 10(27), 9573.
View at Publisher | View at Google Scholar - Koffas, A., Giakoustidis, A., Papaefthymiou, A., Bangeas, P., Giakoustidis, D., Papadopoulos, V. N., & Toumpanakis, C. (2023). Diagnostic work- up and advancement in the diagnosis of gastroenteropancreatic
View at Publisher | View at Google Scholar - Bonestroo, L., & Racila, E. (2023). Histopathologic Diagnosis of Neuroendocrine Neoplasms of Head and Neck, Lung and Gastrointestinal Tract.
View at Publisher | View at Google Scholar - Abbas, F. (2022) “HISTOPATHOLOGICAL PROFILE OF GASTROINTESTINAL NEUROENDOCRINE TUMORS IN A TERTIARY CARE HOSPITAL”, International Journal of Medicine and Medical Research, 7(2), pp. 83–90. doi: 10.11603/ijmmr.2413- 6077.2021.2.12595.
View at Publisher | View at Google Scholar - Öberg, K., Califano, A., Strosberg, J. R., Ma, S., Pape, U., Bodei, L., ... & Paulson, S. (2020). A meta-analysis of the accuracy of a neuroendocrine tumor mRNA genomic biomarker (NETest) in blood. Annals of Oncology, 31(2), 202-212.
View at Publisher | View at Google Scholar - Van Treijen, M. J., Korse, C. M., Van Leeuwaarde, R. S., Saveur, L. J., Vriens, M. R., Verbeek, W. H., ... & Valk, G. D. (2018). Blood transcript profiling for the detection of neuroendocrine tumors: results of a large independent validation study. Frontiers in endocrinology, 9, 740.
View at Publisher | View at Google Scholar - Hennrich U, Benešová M. [68Ga] Ga-DOTA- TOC: The First FDA-Approved 68Ga-Ra- diopharmaceutical for PET Imaging. Pharmaceuticals (Basel). 2020 Mar 3;13(3):38. doi: 10.3390/ph13030038. PMID: 32138377; PMCID: PMC7151717.
View at Publisher | View at Google Scholar - Yu, J., Cao, F., Zhao, X., Xie, Q., Lu, M., Li, J., ... & Sun, Y. (2022). Correlation and comparison of somatostatin receptor type 2 immunohistochemical scoring systems with 68Ga-DOTATATE positron emission tomography/computed tomography imaging in gastroenteropancreaticneuroendocrine neoplasms. Neuroendocrinology, 112(4), 358-369.
View at Publisher | View at Google Scholar - Kunikowska, J., Ambrosini, V., & Herrmann, K. (2021). EANM Focus 3: The International Conference on Molecular Imaging and Theranostics in Neuroendocrine Tumours—the consensus in a nutshell. European Journal of Nuclear Medicine and Molecular Imaging, 48(5), 1276-1277.
View at Publisher | View at Google Scholar - Treglia, G., Sadeghi, R., Giovinazzo, F., Galiandro, F., Annunziata, S., Muoio, B., & Kroiss, A. S. (2021). PET with different radiopharmaceuticals in neuroendocrine neoplasms: An umbrella review of published meta-analyses. Cancers, 13(20), 5172.
View at Publisher | View at Google Scholar - Piccardo, A., Fiz, F., Bottoni, G., Ugolini, M., Noordzij, W., & Trimboli, P. (2021). Head‐to‐head comparison between 18F‐DOPA PET/CT and 68Ga‐DOTA peptides PET/CT in detecting intestinal neuroendocrine tumours: A systematic review and meta‐analysis. Clinical endocrinology, 95(4), 595-605.
View at Publisher | View at Google Scholar - Alevroudis, E., Spei, M. E., Chatziioannou, S. N., Tsoli, M., Wallin, G., Kaltsas, G., & Daskalakis, K. (2021). Clinical utility of 18F-FDG PET in neuroendocrine tumors prior to peptide receptor radionuclide therapy: A systematic review and meta-analysis. Cancers, 13(8), 1813.
View at Publisher | View at Google Scholar - Fang JM, Li J, Shi J. An update on the diagnosis of gastroenteropancreatic neuroendo- crine neoplasms. World J Gastroenterol. 2022 Mar 14;28(10):1009-1023. doi: 10.3748/wjg. v28.i10.1009. PMID: 35431496; PMCID: PMC8968521.
View at Publisher | View at Google Scholar - Gherghe, M., Lazăr, A. M., Stanciu, A. E., Mutuleanu, M. D., Sterea, M. C., Petroiu, C., & Galeș, L. N. (2022). The New Radiolabeled Peptide 99mTcEDDA/HYNIC-TOC: Is It a Feasible Choice for Diagnosing Gastroenteropancreatic NETs?. Cancers, 14(11), 2725.
View at Publisher | View at Google Scholar - Vahidfar, N., Farzanehfar, S., Abbasi, M., Mirzaei, S., Delpassand, E. S., Abbaspour, F., ... & Ahmadzadehfar, H. (2022). Diagnostic value of radiolabelled somatostatin analogues for neuroendocrine tumour diagnosis: the benefits and drawbacks of [64Cu] Cu-DOTA- TOC. Cancers, 14(8), 1914.
View at Publisher | View at Google Scholar - Inaba, Y., Hijioka, S., Iwama, I., Asai, T., Miyamura, H., Chatani, S., ... & Hara, K. (2022). Clinical usefulness of Somatostatin Receptor Scintigraphy in the Diagnosis of Neuroendocrine Neoplasms. Asia Oceania Journal of Nuclear Medicine and Biology, 10(1), 1.
View at Publisher | View at Google Scholar - Yachida, S., Totoki, Y., Noë, M., Nakatani, Y., Horie, M., Kawasaki, K., ... & Shibata, T. (2022). Comprehensive genomic profiling of neuroendocrine carcinomas of the gastrointestinal system. Cancer discovery, 12(3), 692-711.
View at Publisher | View at Google Scholar - Takayanagi, D., Cho, H., Machida, E., Kawamura, A., Takashima, A., Wada, S., ... & Shiraishi, K. (2022). Update on epidemiology, diagnosis, and biomarkers in gastroenteropancreatic neuroendocrine neoplasms. Cancers, 14(5), 1119.
View at Publisher | View at Google Scholar - Doroudinia, A., Emami, H., & Hosseini, M. S. (2022). 68Ga-DOTATATE Radioisotope scan to detect neuroendocrine tumors; A Cross- Sectional Study. Asia Oceania Journal of Nuclear Medicine and Biology, 10(1), 14.
View at Publisher | View at Google Scholar - Canakis, A., & Lee, L. S. (2022). Current updates and future directions in diagnosis and management of gastroenteropancreatic neuroendocrine neoplasms. World journal of gastrointestinal endoscopy, 14(5), 267.
View at Publisher | View at Google Scholar - Hofland, J., Kaltsas, G., & de Herder, W. W. (2020). Advances in the diagnosis and management of well-differentiated neuroendocrine neoplasms. Endocrine Reviews, 41(2), 371-403.
View at Publisher | View at Google Scholar - Öberg, K. (2021). Molecular genomic blood biomarkers for neuroendocrine tumors: the long and winding road from Berzelius and Bence Jones to a neuroendocrine destination. Neuroendocrinology, 111(4), 297- 303.
View at Publisher | View at Google Scholar - Walczyk, J., & Sowa-Staszczak, A. (2019). Diagnostic imaging of gastrointestinal neuroendocrine neoplasms with a focus on ultrasound. Journal of ultrasonography, 19(78), 228-235.
View at Publisher | View at Google Scholar - Ahmed, M. (2020). Gastrointestinal neuroendocrine tumors in 2020. World journal of gastrointestinal oncology, 12(8), 791.
View at Publisher | View at Google Scholar - Dai, M., Mullins, C. S., Lu, L., Alsfasser, G., & Linnebacher, M. (2022). Recent advances in diagnosis and treatment of gastroenteropancreatic neuroendocrine neoplasms. World Journal of Gastrointestinal Surgery, 14(5), 383.
View at Publisher | View at Google Scholar - Shah, M. H., Goldner, W. S., Benson, A. B., Bergsland, E., Blaszkowsky, L. S., Brock, P., ... & Hochstetler, C. (2021). Neuroendocrine and adrenal tumors, version 2.2021, NCCN clinical practice guidelines in oncology. Journal of the National Comprehensive Cancer Network, 19(7), 839-868.
View at Publisher | View at Google Scholar - La Rosa, S., & Uccella, S. (2021). Classification of neuroendocrine neoplasms: lights and shadows. Reviews in endocrine and metabolic disorders, 22(3), 527-538.
View at Publisher | View at Google Scholar - Ariani, G. D. W., & Faruk, M. (2020). Colorectal neuroendocrine tumors: a case series. International Journal of Surgery Case Reports, 72, 411-417.
View at Publisher | View at Google Scholar

 Clinic
Clinic
