Research Article | DOI: https://doi.org/10.31579/2835-835X/126
In-Vitro Samples Treatment from Urinary Tract Infections Patients by Saudi Apium graveolens
*Corresponding Author: Sherifa Mostafa M. Sabra S. Const., Prof., Dr. Microbiology.
Citation: Sherifa Mostafa M. Sabra, (2025), In-Vitro Samples Treatment from Urinary Tract Infections Patients by Saudi Apium graveolens, Clinical Trials and Case Studies, 5(2); DOI:10.31579/2835-835X/126
Copyright: © 2025, Sherifa Mostafa M. Sabra. This is an open-access artic le distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 23 March 2026 | Accepted: 06 April 2026 | Published: 17 April 2026
Keywords: apium graveolens l; proteus sp; staphylococcus aureus; klebsiella pneumonia; serratia sp; enterobacter sp; escherichia coli; candida albicans; staphylococcus epidermidis; and streptococcus sp
Abstract
Apium graveolens L is herb and spice widely grown in Saudi Arabia. Apium graveolens had active principles served as a primary basis of effective antimicrobial agents. The goal was to demonstrate Saudi Apium graveolens extract role in eliminating urinary tract infection pathogens and to highlight the importance of consuming medically appropriate for individuals at risk of infection to reduce the incidence of urinary tract infections. Methodology was for testing of Saudi Apium graveolens crude extract against urinary tract infection pathogens. The results revealed the urinary tract infection pathogens were Proteus sp., Staphylococcus aureus, Klebsiella pneumonia, Serratia sp., Enterobacter sp., Escherichia coli, Candida albicans, Staphylococcus epidermidis, and Streptococcus sp. The first urinary tract infection pathogen was Proteus sp., the partial eradication was at 24 hours 1.7%, and was complete eradication 0.00% after 36 hours. The second group were Staphylococcus aureus, Klebsiella pneumonia, and Serratia sp. Partial eradication was after 24 hours were 1.9%, 1.5%, and 1.3%, and complete eradication after 36 hours were 0.00%, 0.00%, and 0.00%. The third group were Enterobacter sp., Escherichia coli, and Candida albicans. Partial eradication was after 36 hours were 1.8%, 1.2%, and 0.5%, and complete eradication after 48 hours were 0.00%, 0.00%, and 0.00%. The fourth group were Staphylococcus epidermidis, and Streptococcus sp. Partial eradication after 48 hours were 1.6%, and 1.7%. That concluded Saudi Apium graveolens extract was capable of eliminating urinary tract infection pathogens. That recommend the using a medically appropriate Saudi Apium graveolens extract to reduce the incidence of urinary tract infection and accelerate their treatment.
Introduction
Apium graveolens L is a well-known fragrant herb and spice that is widely grown in Saudi Arabia. Raw, cooked, or processed is used in food. Apium graveolens traditionally, the whole plant was consumed for the treatment of genitourinary treatments [1]. Apium graveolens had been used in traditional medicine as a diuretic for bladder and kidney complaint [2]. Apium graveolens essential oils were (terpenes, phthalides, and aldehydes) contribute to the distinctive odor and flavor [3]. Apium graveolens had a variety of phytochemicals, the most important of which are limonene, phthalides, β-salinene, coumarins, spathul-enol, and flavonoids (apiin), which were the primary constituents of essential oils [4]. Apium graveolens had a fresh and strong smell to the high concentration of limonene, both the enantiomer (R)-(+)-limonene with a citrusy odor and the enantiomer (S)-(−)-limonene with a pine-fresh odour [5].
Apium graveolens had nonsteroidal anti-inflammatory drugs (NSAIDs) such as ibuprofen, diclofenac, aspirin, and naproxen were anti-inflammatory drugs currently used for inflammation treatments [6]. Apium graveolens had active principles served as a primary basis of effective antimicrobial agents [7]. Antimicrobial activity of Apium graveolens crude extract had bactericidal activity against different Gram-positive, Gram-negative and fungal strains. The extracts and their bioactive constituents were known to have antimicrobial activity [8]. Apium graveolens L were screened for their antimicrobial effects against Gram-positive; Staphylococcus aureus; Gram-negative; Klebsiella pneumonia, and fungal strains; Candida albicans. Apium graveolens demonstrated to possess high inhibition against Gram-positive and Gram-negative strains extended between 12-21 mm and listed fungal strains had 07-21 mm. The value of MIC of A. graveolens L extended to 250-500 µg/ml [9]. Apium graveolens exhibited significant antibacterial activity against Escherichia coli, and Staphylococcus aureus [10].
The goal was to treat samples from infected individuals using a simple Apium graveolens extract to demonstrate its role in eliminating urinary tract infection pathogens and to highlight the importance of consuming medically appropriate simple Apium graveolens extracts for individuals at risk of infection to reduce the incidence of urinary tract infections.
Methodology:
Plant Extract:
Fresh samples of the Saudi Apium graveolens were purchased from the main market. The sample was washed and left to dry for one week. The dried plant was ground using a sterile grinder. Fifty g of powder was mixed in 200 ml of distilled water in "Soxhlet-Apparatus" for 24 hours. The mixture was filtered by filter paper "Whatman No.1" and the filtrate was concentrated in a rotary evaporator. The concentrated crude extract was stored at 4 °C [11].
Sterile Samples:
Sterile Samples of urine were collected from patients with urinary tract infections from "Micro. Lab.". Samples were prepared by sterile precipitation [12].
Anti-Urinary Tract Infection Pathogens Method:
The sterile precipitate was direct cultured onto "Molar-Hinton-Agar"; (zero culture) and the pathogen colonies were isolated and identified. Equal amounts were placed of sterile precipitate and Saudi Apium graveolens crude extract in "Wassermann Tubes" with good mix, the tube was incubated at 37 °C. The tube content was filtered every twelve hours and was inoculated the precipitate sheet onto "Molar-Hinton-Agar", the resulting the pathogen colonies percent were calculated by equation "Colony count/300 X 100" [13].
Statistical Analyses
The results were giving triple trials, were existed as mean with standard errors. These final results were intended by "Microsoft-Excel-Software" [14].
Results and Discussions:
Isolated pathogens | Time | ||||
Zero hours | 12 hours | 24 hours | 36 hours | 48 hours | |
Proteus sp. | 91.8% | 15.2% | 1.7% | 0.00% | 0.00% |
|
|
|
|
|
|
Staphylococcus aureus | 92.1% | 26.5% | 1.9% | 0.00% | 0.00% |
Klebsiella pneumonia | 91.5% | 23.4% | 1.5% | 0.00% | 0.00% |
Serratia sp. | 92.5% | 24.9% | 1.3% | 0.00% | 0.00% |
|
|
|
|
|
|
Enterobacter sp. | 92.2% | 56.2% | 26.3% | 1.8% | 0.00% |
Escherichia coli | 91.8% | 59.3% | 28.1% | 1.2% | 0.00% |
Candida albicans | 81.2% | 32.3% | 19.7% | 0.5% | 0.00% |
|
|
|
|
|
|
Staphylococcus epidermidis | 93.1% | 66.3% | 42.2% | 21.3% | 1.6% |
Streptococcus sp. | 91.3% | 68.2% | 41.3% | 23.4% | 1.7% |
Table 1: Average mean for urinary tract infection pathogens eradication after exposure to Saudi Apium graveolens extract
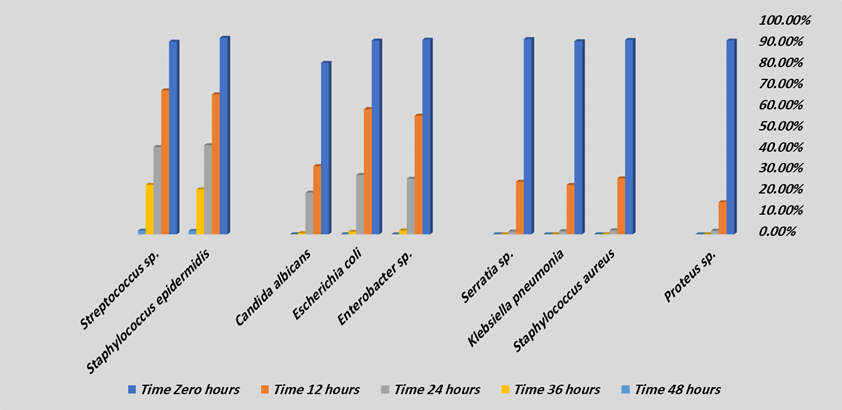
Figure 1: Average mean for urinary tract infection pathogens eradication after exposure to Saudi Apium graveolens extract
Table (1), and Figure (1) were showed average mean for urinary tract infection pathogens eradication after exposure to Saudi Apium graveolens extract. In the first step, the following isolates were isolated and identified as urinary tract infection pathogens were included Proteus sp., Staphylococcus aureus, Klebsiella pneumonia, Serratia sp., Enterobacter sp., Escherichia coli, Candida albicans, Staphylococcus epidermidis, and Streptococcus sp. After exposure to Saudi Apium graveolens extract, the urinary tract infection pathogens were tested to determine their partial or complete eradication. Each isolate pathogen was exposed for 48 hours, and microbial growth was monitored to assess the degree of microbial eradication after exposure to Saudi Apium graveolens extract. The
experimental results depended on the extract's effect on the isolated pathogens obtained from sterile urine samples [6-10].
The experiment was depending on the chemical content of Saudi Apium graveolens extract as antimicrobial [1-8].
The initial result for the first urinary tract infection pathogen was Proteus sp., the partial eradication was at 24 hours as 1.7%, was followed by complete eradication 0.00?ter 36 hours [6-10].
The second group of the urinary tract infection pathogens were included Staphylococcus aureus, Klebsiella pneumonia, and Serratia sp. Partial eradication was occurred after 24 hours were (1.9%, 1.5%, and 1.3%), were followed by complete eradication after 36 hours were (0.00%, 0.00%, and 0.00%) [6-10].
The third group of the urinary tract infection pathogens were included Enterobacter sp., Escherichia coli, and Candida albicans. Partial eradication was occurred after 36 hours were (1.8%, 1.2%, and 0.5%), were followed by complete eradication after 48 hours were (0.00%, 0.00%, and 0.00%) [6-10].
The fourth group of the urinary tract infection pathogens were included Staphylococcus epidermidis, and Streptococcus sp. Partial eradication was occurred after 48 hours were (1.6%, and 1.7%), so this in need more one day in time for complete eradication [6-10].
The results showed that Saudi Apium graveolens extract contains active ingredients that eliminate Gram-positive, Gram-negative bacteria and fungi that cause urinary tract infection. The time required for elimination of urinary tract infection pathogens may vary depending on the type of isolate and the concentration of the Saudi Apium graveolens extract [1-8].
Conclusions:
That concluded the Saudi Apium graveolens extract was capable of eliminating urinary tract infection pathogens.
Recommendations:
That recommend the using a medically appropriate Saudi Apium graveolens extract to reduce the incidence of urinary tract infection and accelerate their treatment.
Acknowledgments:
The extend sincere thanks to the research collaborator.
References
- Wahba, M., Abdein, A., Alhaithloul, A., Alghanem, M., et al. (2025). Advances in Dill (Anethum graveolens L.) Molecular Breeding Strategies. In Biodiversity and Genetic Improvement of Herbs and Spices (pp. 89-120). Cham: Springer Nature Switzerland.
View at Publisher | View at Google Scholar - Mohammed, A. et al. (2025). Traditional Medicinal Plants for Kidney Disease. EC Orthopaedics, 16, 01-09.
View at Publisher | View at Google Scholar - Sun, Y., Li, M., Li, X., Du, J., Li, W., et al. (2023). Characterization of volatile organic compounds in five celery (Apium graveolens L.) cultivars with different petiole colors by HS-SPME-GC-MS. International journal of molecular sciences, 24(17), 13343.
View at Publisher | View at Google Scholar - Foudah, I., Alqarni, H., Alam, A., Salkini, A., Alam, P., et al. (2021). Determination of chemical composition, in vitro and in silico evaluation of essential oil from leaves of Apium graveolens grown in Saudi Arabia. Molecules, 26(23), 7372.
View at Publisher | View at Google Scholar - Wanna, R., Bunphan, D., Kunlanit, B., Khaengkhan, P., Khaengkhan, P., et al. (2025). Chemical composition of essential oil from Apium graveolens L. and its biological activities against Sitophilus zeamais Motschulsky (Coleoptera: Dryophthoridae). Plants, 14(3), 347.
View at Publisher | View at Google Scholar - Abu-Baih, H., Gomaa, R., Abdel-Wahab, M., Abdelaleem, R., Zaher, A., et al. (2024). Apium extract alleviates indomethacin-induced gastric ulcers in rats via modulating the VEGF and IK-κB/NF-κB p65 signaling pathway: insights from in silico and in vivo investigations. BMC Complementary Medicine and Therapies, 24(1), 88.
View at Publisher | View at Google Scholar - Zhang, Y., Wang, T., Wang, X., Dong, R., Wang, D., et al. (2025). Apium Graveolens Linn. (celery) seeds: A comprehensive review of phytochemistry profile, pharmacological effects and future trends. Food Reviews International, 1-28.
View at Publisher | View at Google Scholar - Emad, M., Rasheed, M., El-Kased, F., & El-Kersh, M. et al. (2022). Antioxidant, antimicrobial activities and characterization of polyphenol-enriched extract of Egyptian celery (Apium graveolens L., Apiaceae) aerial parts via UPLC/ESI/TOF-MS. Molecules, 27(3), 698.
View at Publisher | View at Google Scholar - Bushra, R., Saeed, F., Ahmed, Z., Naeem, S., Ishaq, J., et al. (2025). Curbing anti-microbial resistance of synthetic medicinal agents using herbal drug alternatives: Current trends and future insights. Prospects in Pharmaceutical Sciences, 23(2), 50-66.
View at Publisher | View at Google Scholar - sufa, i., kurniati, s., dermawan, a., abror, k., indra, n., et al. (2024). Therapeutic potential of multi-targeting phytochemicals derived from Apium graveolens ethanol extract in West Java, Indonesia against multidrug-resistant Pseudomonas aeruginosa. Biodiversitas: Journal of Biological Diversity, 25(5).
View at Publisher | View at Google Scholar - Singh, M., Rajput, M., Yadav, K., & Singh, N. et al. (2022). Evaluation of antimicrobial activity and phytochemical qualitative analysis of Ephedra foliata Boiss. ex CA Mey. Herba Polonica, 68(2), 69-75.
View at Publisher | View at Google Scholar - vinay, h. et al. (2022). diagnosis and management of lower urinary tract affections in dogs (doctoral dissertation, chaudhary sarwan kumar himachal pradesh krishi vishvavidyalaya).
View at Publisher | View at Google Scholar - Otajevwo, F., & Osawaru, E. et al. (2020). Testing the efficacy of Mueller Hinton agar over Nutrient agar for optimal antibiotic sensitivity testing response by selected clinical bacterial pathogens. GSC Adv. Res. Rev, 5, 061-074.
View at Publisher | View at Google Scholar - Kiviaho, K., Anttonen, M., Alfthan, H., & Itkonen, O. ET AL. (2025). Reliable quantification of fecal elastase-1: A study on sample stability, IDK ELISA and IDK Extract® device. Clinica Chimica Acta, 120541.
View at Publisher | View at Google Scholar

 Clinic
Clinic
