Review Article | DOI: https://doi.org/10.31579/2834-8761/104
Breast Cancer: Causes, Molecular Pathogenesis, Tumour Microenvironment Diagnosis, and Emerging Therapeutic Approaches
Department of Zoology, Deen Dayal Upadhyay Gorakhpur University, Gorakhpur, India
*Corresponding Author: Ravi Kant Upadhyay, Department of Zoology, Deen Dayal Upadhyay Gorakhpur University, Gorakhpur, India.
Citation: Apoorva Dubey, Ravi K. Upadhyay, (2026), Breast Cancer: Causes, Molecular Pathogenesis, Tumour Microenvironment Diagnosis, and Emerging Therapeutic Approaches, Clinical Endocrinology and Metabolism, 5(1); Doi:10.31579/2834-8761/104
Copyright: © 2026, Ravi Kant Upadhyay. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Received: 28 January 2026 | Accepted: 11 February 2026 | Published: 17 February 2026
Keywords: breast cancer; epidemiology; markers; tumour microenvironment; drugs; delivery methods; immunotherapy
Abstract
The present article discusses the causes of breast cancer, its diagnosis and control. Breast cancer is a leading cause of death in young women around the globe. This paper aims to diagnose cancer by using imaging, tissue biopsy, and physical examination at an earlier stage. It discusses modern treatment methods, i.e., hormonal therapy, radiation, chemotherapy, surgery, and, more recently, immunotherapy. Variables like histology, stage, tumour markers, and genetic disorders influence individualised therapy choices. It also highlighted key risk factors, including lifestyle changes, delayed marriage, early menstruation, high-energy diets, environmental influences, hormonal factors, and BRCA gene mutations. Most breast cancers arise from luminal epithelial cells of terminal ductal–lobular units and evolve through accumulating somatic alterations under endocrine and microenvironmental selection. This article describes the diagnostics, prevention and control of breast cancer. It also sketches out the use of molecular and genetic markers of breast cancer. It also emphasizes the need for the delivery of targeted medicine by using nanoparticles. It also advocates the use of the antibody-drug conjugation system. There is a need for the development of less toxic and stable drugs or biomolecules compared to traditional chemotherapeutic drugs.
1.Introduction
Breast cancer is the most commonly diagnosed cancer worldwide and a leading cause of cancer related mortality among women, accounting for millions of new cases and hundreds of thousands of deaths each year. Despite improved survival rates in high-income countries, outcomes remain poorer in low and middle-income regions, underscoring disparities in early detection and access to effective treatment. Global incidence continues to rise, with projections suggesting a substantial increase in both cases and deaths by 2040. In 2022 alone, approximately 2.3 million new cases and 670,000 deaths were reported worldwide. Although initiatives such as the WHO Global Breast Cancer Initiative aim to reduce mortality, progress remains limited [1]. Breast cancer encompasses a heterogeneous group of diseases with diverse biological behaviours, prognoses, and therapeutic responses. The most common subtype is invasive ductal carcinoma, originating in the ductal epithelium, followed by lobular carcinoma and other forms such as triple-negative and HER2-positive breast cancers [2]. Clinical presentation often includes a palpable lump or abnormal nipple discharge, particularly in developing regions. Diagnosis relies on physical examination, imaging techniques, and histopathological evaluation through biopsy. Treatment strategies include surgery, chemotherapy, radiotherapy, hormonal therapy, and, more recently, immunotherapy, with therapeutic decisions guided by tumour stage, histology, receptor status, and genetic factors [3]. The development of breast cancer is influenced by a combination of genetic, hormonal, environmental, and lifestyle related factors, including BRCA gene mutations, early menarche, delayed childbearing, dietary habits, and environmental exposures. Anatomically, the breast consists of lobules, ducts, adipose tissue, and supporting structures such as Cooper’s ligaments, all of which play a role in disease origin and progression. Given its complex aetiology and heterogeneity, breast cancer requires a multifaceted and personalized approach to management. This review aims to highlight the key mechanisms underlying breast cancer progression, along with current diagnostic, therapeutic, and long-term management strategies [4-5].
2. Source of data
For writing this comprehensive research review on breast cancer were searched. An extensive search was conducted to explore our research outcomes on breast cancer from 1999 onwards. The articles were thoroughly read for recent research in this area. For relevant information, literature electronic databases were searched, using keywords, and specific terms such as Infectivity, disease transmission and pathogenesis, medical subject headings (MeSH) and key text words, such as breast cancer, tumour microenvironment, metastasis, types of breast cancer, pathogenesis, mRNA vaccines and its control till 2025 were used in MEDLINE. For various database searches, the emphasis was given to collect scientific information. The topic was searched very widely to collect the maximum information on the topic “Breast Cancer: causes, pathogenesis, diagnosis, and therapeutics”.Papers were selected on the basis of work done related to breast cancer, and a systematic approach was applied to organize the available information on all facets of vaccine development. Data searches were done on the key points’ invasion of host tissues and cells, immune responses, and mechanisms of pathogenesis, symptoms, control measures and precautions. From a study point of view, the literature available on the genetic basis of cancer development and mutagens that increase cancer risk. However, glaring gaps in knowledge of many critical subjects, such as immunotherapy, stem cell therapy, and vaccine development, were preferentially selected.
3. Epidemiology:
Breast cancer is the most commonly diagnosed cancer in women worldwide and a major cause of cancer-related deaths. According to the International Agency for Research on Cancer (IARC) GLOBOCAN 2022, about 2.3 million new cases and 666,000 deaths were reported globally, accounting for a significant share of the overall cancer burden. It represents nearly one in four cancer cases in women, underscoring its major public health impact [6]. The incidence of breast cancer varies across regions, with global trends showing a steady rise over recent decades. This increase is largely driven by population ageing, urbanisation, and lifestyle changes such as altered reproductive patterns and obesity. Age is a key risk factor, with most cases occurring in women over 40 years and peaking between 50 and 60 years [7]. Although advances in early detection and treatment have improved survival, particularly in high-income countries, marked disparities remain. These differences highlight the need for effective screening strategies and improved access to healthcare to reduce the global burden of breast cancer [8].
4. Risk factors:
Breast cancer is influenced by various factors, with higher incidence rates in middle-aged women. Ductal or lobular carcinoma in situ increases recurrence risk. Women with dense breast tissue are more susceptible to developing breast cancer. Menopause affects hormone levels and risk; later menopause is linked to increased risk, while early menopause decreases it. Pregnancy alters breast cell exposure to estrogen and reduces menstrual cycles; having children generally lowers breast cancer risk, whereas nulliparity raises it. The incidence rate is high in middle-aged women (Ait Zenati et al, 2025). Fatty tissue appears black, hiding a tumour, whereas dense tissue appears white, resembling malignancies [9]. During pregnancy, breast cells stop being exposed to estrogen, reducing the number of menstrual cycles. Having one full-term pregnancy before 30 slightly raises breast cancer risk, while early pregnancy and having more children lower that risk. Not conceiving increases the risk [10].
4.1 Level of Estrogens:
Both endogenous and exogenous estrogens are associated with an increased risk of breast cancer. Endogenous estrogens are normally produced by the ovary in premenopausal women, and ovarian excision can reduce the risk of breast cancer. The two main sources of exogenous estrogen are oral contraceptives and hormone replacement therapy. Oral contraceptives have been widely used since the 1960s, and formulas have been refined to reduce adverse effects [11].
4.2 Lifestyle:
Lifestyle factors such as radiation exposure, obesity, physical inactivity, alcohol consumption, and smoking increase the risk of breast cancer. Recent research also suggests that disruptions in circadian rhythms may contribute to cancer development by altering cell cycle regulation and reducing melatonin secretion [12]. In contrast, regular physical activity may lower risk by modulating hormonal balance, enhancing immune function, and reducing inflammation, whereas circadian disturbances may promote oncogenesis through decreased melatonin levels and heightened inflammatory responses [13].
4.3 Being overweight:
Obesity is a significant risk factor for breast cancer, particularly among overweight postmenopausal women. Estrogens, especially those associated with ovulation, plays a key role, and prolonged cumulative exposure to estrogen in breast tissue contributes to increased risk. Excess adipose tissue elevates estrogen levels, further promoting disease development. While the link between overall dietary fat and breast cancer is well established, the role of dietary fat quality indices remains less clearly understood [14].
4.4 Genetic reasons:
When several close blood relatives have breast cancer, it suggests the disease may run in their family. Some families have more cases of breast cancer than usual, often without meaning to. It can be hard to figure out if this family history is just by chance, due to common lifestyle choices, inherited genes, or a combination of these factors [15].
4.5 Gene mutations in the BRCA
A genetic mutation is an alteration in a gene that can increase the risk of certain cancers, and some of these mutations can be inherited from parents. The BRCA1 and BRCA2 genes normally help protect against cancer, but mutations in these genes can impair this function. Although relatively rare, such mutations are associated with a significantly increased cancer risk [16].
5. Genes responsible for breast cancer:
Inherited mutations in the BRCA1 and BRCA2 genes are the most common contributors to hereditary breast cancer, markedly increasing lifetime risk. These tumour suppressor genes normally function in DNA repair, but mutations impair this process, resulting in uncontrolled cell proliferation. Other important high-risk genes include PALB2, TP53, PTEN, CHEK2, and CDH1 [17]. BRCA1, located on chromosome 17, was the first key gene identified, while PALB2 is now the third most common gene associated with breast cancer, working alongside BRCA2 to repair DNA. Women with PALB2 mutations have a higher chance of developing breast cancer by age 70. CHEK2, or "Checkpoint Kinase 2," produces a protein that helps stop tumour growth. Women with a CHEK2 gene mutation are twice as likely to develop breast cancer, while men with the mutation have a tenfold increased risk of breast cancer [18]. CDH1, or "CaDHerin 1," is a gene that helps cells stick together to form organized tissues. A mutation in the CDH1 gene can raise the risk of lobular breast cancer and may allow cancer cells to break away from a tumour and spread to other body parts [19]. The PTEN gene helps stop tumour growth by managing cell division and encouraging damaged cells to die. It also aids in cell adhesion, which may help prevent cancer from spreading. STK11 is another gene that can suppress tumours and is associated with Peutz-Jeghers syndrome, increasing the risk of breast cancer. TP53, known as P53, acts as a protector of genetic material by spotting DNA damage and either urging cell death or repair. Mutations in TP53, whether inherited or developed over time, disrupt DNA repair processes, which can lead to cancer [20-21].
6. Molecular classification of breast cancer:
Breast cancer, a biologically and clinically diverse disease comprise multiple distinct subtypes, and it’s classify cation has evolved considerably over time. The most widely accepted system is based on immunohistochemical profiling, which evaluates the expression of key receptors, including estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2) [22]. Based on these markers, breast cancer is broadly categorised into four major subtypes: luminal A, luminal B, HER2-positive, and triple negative [23]. Breast tumours with estrogen receptor (ER) expression of ≥1% are classified as luminal A subtype. These tumours typically exhibit high progesterone receptor (PR) [removed]≥20%) and low Ki-67 levels (<20>10%). In HER2-negative luminal B tumours, progesterone receptor (PR) expression is typically low or absent (<20>
7. Pathogenesis:
Most breast cancers start from luminal epithelial cells and develop through genetic changes influenced by hormones and their surroundings. In recent years, oncology research has increasingly focused on developing targeted therapies aimed at specific molecular alterations in breast cancer [27]. These approaches include monoclonal antibodies, tyrosine kinase inhibitors, antibody drug conjugates, inhibitors of the PI3K/Akt/mTOR pathway, CDK4/6 inhibitors, PARP inhibitors, anti-angiogenic agents, and other targeted drugs. In addition, immunomodulatory strategies such as immune checkpoint inhibitors (anti-PD-1/PD-L1), CTLA-4 inhibitors, adoptive T-cell therapy, and cancer vaccines are designed to enhance the immune system’s ability to recognize and eliminate tumour cells. Epigenetic therapies, including DNA methyltransferase (DNMT) and histone deacetylase (HDAC) inhibitors, have also demonstrated promising potential in breast cancer treatment [28]. Breast cancer arises through a multistep process involving the accumulation of genetic and epigenetic alterations in mammary epithelial cells, leading to dysregulated cell proliferation, survival, and differentiation. Key driver events include mutations in oncogenes and tumor suppressor genes such as PIK3CA, TP53, and BRCA1/2, which contribute to genomic instability and impaired DNA repair mechanisms [29]. Aberrant activation of signalling pathways plays a central role, particularly the PI3K/AKT/mTOR and MAPK pathways, which promote tumour growth, survival, and resistance to apoptosis. Amplification of the HER2/ERBB2 gene further enhances proliferative and metastatic signalling, defining an aggressive molecular subtype of breast cancer [30]. Epigenetic modifications, including DNA methylation and histone alterations, regulate gene expression and contribute to oncogenesis by silencing tumor suppressor genes and activating oncogenic pathways. Hormonal signaling, especially through estrogen receptor pathways, also drives tumour initiation and progression in hormone receptor positive breast cancers [31]. The tumour microenvironment plays a critical role in disease progression by facilitating angiogenesis, immune evasion, and metastasis through interactions between cancer cells, stromal cells, and immune components. Metastatic spread is closely associated with epithelial-mesenchymal transition (EMT), enabling tumour cells to acquire invasive and migratory capabilities. Additionally, immune evasion via checkpoint molecules such as PD-L1 and the development of tumor cell dormancy contribute to disease persistence and recurrence [32]. These molecular alterations collectively drive breast cancer heterogeneity, progression, and therapeutic resistance.
8. Tumour microenvironment of breast cancer:
The tumour microenvironment (TME) is a dynamic network of cancer cells, stromal components, immune cells, and non-cellular elements that collectively regulate tumour progression, metastasis, and therapeutic response. Stromal components, particularly cancer-associated fibroblasts (CAFs) and adipocytes, promote tumorigenesis through extracellular matrix (ECM) remodelling, angiogenesis, immune suppression, and therapy resistance. CAFs secrete cytokines such as IL-6, TGF-β, FGF, and HGF, along with enzymes like matrix metalloproteinases (MMPs) and lysyl oxidase (LOX), which enhance ECM stiffness and limit drug penetration. In triple-negative breast cancer, specific CAF subtypes are associated with immunosuppressive signalling and reduced CD8⁺ T-cell activity [33]. Immune cells within the TME exhibit dual roles. Tumour-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs) promote angiogenesis, metastasis, and immune evasion, whereas tumour infiltrating lymphocytes (TILs) may contribute to either tumor suppression or progression depending on their composition [34]. Non-cellular components, including hypoxia, ECM stiffness, acidic pH, and exosomes, further influence tumour behaviour. Hypoxia-driven HIF-1α signaling promotes epithelial-mesenchymal transition, stemness, and therapeutic resistance, while exosomes facilitate intercellular communication and metastasis. Overall, these findings highlight how complex the breast TME is, acting as both a promoter of tumour growth and a target for treatment. Targeting the TME alongside conventional and targeted therapies could help overcome resistance and enhance treatment success.
9. Tumour microenvironment in therapy resistance:
The tumour microenvironment (TME) plays a crucial role in therapy resistance in breast cancer by creating a protective niche that supports tumour survival and limits drug efficacy. Cancer-associated fibroblasts (CAFs) promote resistance through secretion of cytokines such as TGF-β, IL-6, and HGF, which activate survival pathways including PI3K/AKT and MAPK signalling. Additionally, extracellular matrix (ECM) remodelling by matrix metalloproteinases (MMPs) and lysyl oxidase (LOX) reduces drug penetration and enhances tumour progression [35]. Immune components further contribute to resistance. Tumour-associated macrophages (TAMs) and myeloid-derived suppressor cells (MDSCs) suppress cytotoxic T-cell activity and promote immune evasion via immunosuppressive cytokines and checkpoint molecules such as PD-L1, thereby reducing the effectiveness of immunotherapy [36]. Hypoxia within the TME activates HIF-1α signalling, inducing epithelial–mesenchymal transition (EMT), metabolic reprogramming, and expression of drug resistance genes. Moreover, extracellular vesicles, particularly exosomes, facilitate the transfer of proteins and microRNAs that enhance drug resistance and metastatic potential [37].
10. Diagnosis:
The diagnosis of breast cancer involves a multimodal approach integrating clinical evaluation, imaging techniques, and pathological as well as molecular analyses. Early and accurate diagnosis is essential for improving prognosis and guiding personalized therapy. Initial assessment includes physical and clinical examination, where palpable masses, nipple discharge, or skin changes are evaluated. However, definitive diagnosis relies on diagnostic imaging, particularly mammography, ultrasound, and magnetic resonance imaging (MRI) [38]. Mammography remains the gold standard for screening and early detection, while ultrasound is useful in differentiating solid from cystic lesions and is commonly used in younger women. MRI provides high sensitivity and is recommended for high risk [39]. Confirmation of malignancy requires histopathological evaluation through biopsy techniques, including fine needle aspiration (FNA), core needle biopsy, and surgical biopsy. Among these, core needle biopsy is widely preferred due to its accuracy and ability to preserve tissue architecture for further analysis. In addition, laboratory and genomic testing play a crucial role in characterization of tumours (figure.1). Determination of hormone receptor status (estrogen receptor [ER] and progesterone receptor [PR]) and human epidermal growth factor receptor 2 (HER2) status is essential for classification and treatment selection [40]. Advanced genomic assays further assist in prognosis and therapeutic decision-making.
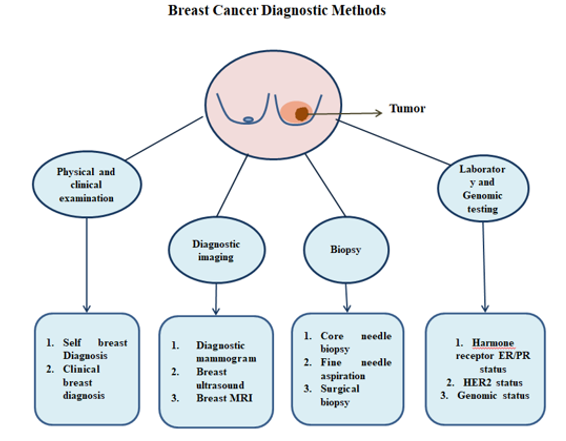
Figure1: Diagnostic Methods Used for Detection of Breast Cancer.
11. Therapeutics:
Breast cancer treatment requires a multidisciplinary approach based on tumor burden and molecular markers. Early-stage disease is managed with breast-conserving surgery plus radiotherapy or mastectomy, with adjuvant therapy guided by lymph node and receptor status. In metastatic cases, treatment focuses on prolonging survival and maintaining quality of life. For non-metastatic breast cancer, surgery is the standard treatment, while preoperative chemotherapy can shrink tumors, enabling breast conservation and reducing the need for axillary lymph node dissection. Advances in endocrine, targeted, and immunotherapies have expanded treatment options for both metastatic and non-metastatic disease (figure.2) [41].
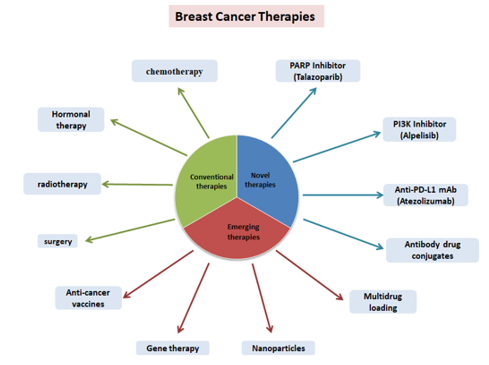
Figure 2: Different therapies used for breast cancer treatment
11.1 Immunotherapy
Immunotherapy for breast cancer leverages the immune system to combat cancer cells, proving vital for specific types like triple-negative and HER2-positive breast cancer. Key treatment modalities include immune checkpoint inhibitors that block proteins inhibiting immune attacks on cancer, and passive immunotherapies, notably monoclonal antibodies targeting specific markers such as those seen in HER2-positive cases [42]. The introduction of pembrolizumab combined with chemotherapy for triple-negative breast cancer (TNBC) marks a significant advancement, integrating immunotherapy into the breast cancer treatment landscape. This progress encourages further research on immune checkpoint inhibitors for other breast cancer subtypes. Clinical trials demonstrate the effectiveness of PD-1/PD-L1 inhibitors with chemotherapy in treating TNBC, leading to their approval and expanding therapeutic options for challenging patient demographics (table.1) [43].
11.2 Endocrine therapy:
Endocrine therapy for breast cancer is standard as adjuvant therapy for HR-positive patients over 5–10 years, with sensitivity linked to hormone receptor expression. In premenopausal patients, a 5-year tamoxifen regimen reduces recurrence risk significantly, with longer duration further decreasing recurrence and mortality. The ATLAS trial suggests a 10-year tamoxifen duration, lowering breast cancer deaths. For patients at high relapse risk, ovarian suppression combined with an aromatase inhibitor (AI) or tamoxifen improves disease-free survival (DFS), though with increased toxicity. In postmenopausal patients, AI monotherapy or sequential treatments are preferred, showing reduced mortality versus tamoxifen. However, AIs have
higher bone-related adverse events, making tamoxifen a viable alternative. The duration of therapy requires further evaluation, potentially using multigene assays for guidance. Neoadjuvant endocrine therapy’s value in early ER-positive breast cancer remains unclear, but combining it with chemotherapy has shown.
11.3 Radiation therapy
Radiation therapy (RT) is essential in the treatment of breast cancer, particularly after breast-conserving surgery (BCS). It is indicated in ductal carcinoma in situ (stage 0) to reduce the risk of local recurrence by 50–60%. In early stage (I-II) invasive breast cancer, RT is the standard post-BCS treatment. For elderly (≥70 years) patients with stage I, hormone receptor-positive tumors, hormonal therapy without RT may be an option. Hypofractionated whole breast irradiation (WBI) and accelerated partial breast irradiation are effective alternatives to conventional WBI [44].
11.4 Gene therapy
Gene therapy is emerging as a promising cancer treatment that involves the delivery of genetic material via a vector to modify gene expression. This encompasses techniques such as gene editing, targeting transcription factors, using microRNA, and DNA or RNA vaccinations. microRNA applications in cancer therapies have shown effectiveness in inhibiting breast cancer cell proliferation and development [45]. The miRNA replacement agent MRX34, involving miR-34a, is currently undergoing clinical trials and is anticipated to significantly contribute to breast cancer treatment in the future. Although the body of research on gene therapy for breast cancer is limited, numerous strategies are progressing into clinical trials [46].
Category | Examples | Mechanism/Target | Clinical Status |
Standard Therapies | Tamoxifen, Anastrozole, Letrozole (Aromatase inhibitors) , Fulvestrant | Endocrine therapy (ER/PR targeting) | FDA-approved |
Chemotherapy | Doxorubicin, Cyclophosphamide, Paclitaxel, Docetaxel | Cytotoxic agents interfering with DNA replication and mitosis | FDA-approved |
Targeted Therapies | Trastuzumab(Herceptin) ,Pertuzumab, Lapatinib,T-DM1 (Ado-trastuzumabemtansine) | HER2-targeted monoclonal antibodies & tyrosine kinase inhibitors | FDA-approved (HER2+ BC) |
CDK4/6 Inhibitors | Palbociclib, Ribociclib, Abemaciclib | Inhibit cyclin-dependent kinases 4/6 → cell cycle arrest | FDA-approved (HR+/HER2– BC) |
PI3K/AKT/mTOR Inhibitors | Alpelisib (PI3K inhibitor), Everolimus (mTOR inhibitor) | Block PI3K/AKT/mTORsignaling pathway | FDA-approved for subsets |
PARP Inhibitors | Olaparib, Talazoparib | Exploit synthetic lethality in BRCA1/2-mutated tumors | FDA-approved |
Immunotherapies | Pembrolizumab (anti–PD-1),Atezolizumab (anti–PD-L1) | Immune checkpoint inhibitors | Approved for TNBC (specific settings) |
Therapeutic Vaccines (Experimental) | NeuVax (E75 peptide, HER2-targeted),GP2 vaccine, AE37 (HER2-derived peptide), DNA & dendritic cell vaccines | Stimulate cytotoxic T-cell response against HER2 or tumours-associated antigens | In clinical trials (Phase II/III) |
Prophylactic Vaccines (Experimental) | α-lactalbumin vaccine (for TNBC prevention),- MUC1-based vaccines | Train immune system against self-antigens expressed in tumours | Preclinical / Early clinical stages |
Table1: Overview of therapeutic strategies for breast cancer.
Drugs
Advancements in genomics, proteomics, and computational biology have led to drug repurposing becoming a new method in cancer treatment. This approach uses existing drugs in new ways, which can help create safer and more effective therapies with fewer side effects. In breast cancer treatment, various repurposed drugs like alkylating agents, anthracyclins, antimetabolites, CDK4/6 inhibitors, aromatase inhibitors, mTOR inhibitors, and mitotic inhibitors have been identified. Specific CDK4/6 inhibitors such as palbociclib, ribociclib, and abemaciclib are more effective when used with aromatase inhibitors, leading to better survival rates for patients [47]. Furthermore, combining aromatase inhibitors like exemestane and tamoxifen with HDAC inhibitors such as entinostat and vorinostat has shown strong anticancer effects. However, about 70% of breast cancer cases experience resistance to hormonal therapy, often related to the activation of the PI3K/AKT/mTOR pathways, with the P110 α (PI3KCA) gene being targeted for new treatments [48]. Everolimus, an FDA-approved mTOR inhibitor, has shown good clinical results when paired with exemestane or trastuzumab in HER2-positive and HER2-negative breast cancer patients.
Natural products
Natural compounds, also known as phytochemicals, have health benefits by affecting specific molecular pathways. They can influence genes directly or support processes that help manage metabolism. Research shows that these phytochemicals, found in food, can change epigenetic events and may reverse changes linked to cancer. They work on important factors that control cell growth, which can stop the cell cycle, promote cell death, and reactivate genes that prevent tumours [49]. Aromatase is a key protein that helps convert certain hormones, playing a vital role in making estrogen. Studies have found that some phytochemicals are similar in structure to estrogen and can reduce aromatase activity. These natural products also help prevent cancer by influencing the arachidonic acid pathway and affecting various metabolic enzymes like COXs, PLA2s, and LOXs.
Vaccines
Breast cancer vaccines are under clinical investigation, with both preventive and therapeutic types being explored. Therapeutic vaccines aim to treat existing cancers by leveraging the immune system to target specific proteins like HER2, whereas preventive vaccines prepare the immune system to combat potential cancer cells through the introduction of benign proteins. Despite eliciting durable immune responses intended to eradicate tumour cells, studies have not demonstrated notable advantages for breast cancer vaccines, particularly as trials for DNA, dendritic cell, and peptide-based vaccines have largely proven unsuccessful due to the complexities of breast cancer and immune system diversity [50]. Emerging therapies such as immune checkpoint inhibitors and anti-HER2 monoclonal antibodies show significant promise in breast cancer treatment. mRNA vaccines are particularly promising, as they induce intracellular antigen production and strong immune responses with low risk of T-cell tolerance. They offer personalized, durable immunity with controlled immunogenicity and generally mild, short-lived side effects. Due to their safety, efficacy, rapid development, and scalability, mRNA vaccines are gaining attention as a novel therapeutic approach, with encouraging progress in preclinical and clinical studies. [51].
Vaccine types:
Cancer vaccines are an effective treatment option because they stimulate a long-lasting immune response that will eliminate tumour cells. In studies on the breast cancer vaccine, no appreciable advantages were discovered. A recent study claims that immune checkpoint inhibitors or anti-HER2 monoclonal antibodies may be used in vaccinations. This vaccination strengthens the immune system to fight off breast cancer cells. Clinical trials have been conducted on DNA, dendritic cells, and peptide-based breast cancer vaccines. Studies on the breast cancer vaccine have employed subcutaneous, intramuscular, and intradermal injections [52]. The complexity of the immune system makes it challenging to create cancer vaccines. Given the heterogeneity of breast cancer, there may be a need for different vaccination strategies.
Antibody-Drug Conjugation Systems (ADCs)
Antibody drug conjugates (ADCs) are a new treatment option for solid tumours that use the interaction between antibodies and antigens. They are made of specific monoclonal antibodies linked to powerful cytotoxins with stable chemical connectors. Important methods for connecting these drugs involve changing amino acid parts of antibodies and breaking disulfide bonds to expose cysteine residues [53]. The success of ADCs is dependent on their design, the type of tumour they target, and several pharmacokinetic factors. ADCs work by binding to target antigens, internalizing the complex, and releasing the drug after degradation. An example, trastuzumab-emtansine (T-DM1), has shown positive results in treating HER2-positive breast cancer, reducing toxicity while boosting antitumor immunity [54]. Many ADCs are also currently being tested in clinical evaluations for effectiveness.
Nanoparticles:
Nanoparticles are tiny carriers designed to deliver drugs directly to the tumour site. They can enhance drug solubility and improve targeting. Liposomes are spherical vesicles made of lipid bilayers that can encapsulate both water-soluble and fat-soluble drugs [55]. Nanoparticles offer advantages such as high drug capacity, lower toxicity, and better stability than traditional chemotherapy. They can deliver anticancer agents to tumours for breast cancer diagnosis and treatment through active or passive methods (table.2). NPs also have multifunctional capabilities. Recently, there has been progress in controlling the release of drugs from NPs to improve effectiveness and counteract multidrug resistance (MDR) [56].
Approaches | Type/carrier | Key Features | Applications |
Systemic (Nanocarriers) | Micelles | ↑ solubility, stability, half-life; bypass efflux pumps | Paclitaxel, doxorubicin; HER-2 targeted micelles |
Liposomes | Carry hydrophilic + hydrophobic drugs; PEGylation prolongs circulation | Doxorubicin, paclitaxel; immunoliposomes | |
Polymers (PLGA, PEG, HPMA) | Biodegradable; controlled release; high drug loading | Paclitaxel, cisplatin; HER-2 polymer conjugates | |
Dendrimers | Branched, multivalent; high loading; tunable
| Doxorubicin, siRNA, PDT enhancers | |
Nanogels | Hydrogel-like; sustained release | siRNA + chemo delivery for MDR | |
Inorganic NPs
| Gold, iron oxide, quantum dots; imaging + therapy | GNPs for therapy, SPIO for MRI, QDs for HER-2 imaging | |
Localized Delivery | Nanofibers | Biodegradable scaffold; local release | Curcumin nanofibers in MCF-7 |
Hydrogels | Injectable; controlled local release | Chitosan hydrogels, siRNA carriers | |
Intraductal Injection | Direct drug delivery into breast ducts | 5-FU, estradiol in ductal lesions | |
Receptor-Targeted | HER-2 | Antibody/kinase inhibition | Trastuzumab, pertuzumab, lapatinib |
EGFR | Block signaling with inhibitors/antibodies | Gefitinib, lapatinib; TNBC | |
IGF-IR
| Tyrosine kinase inhibition | Resistant breast cancers | |
VEGFR | Anti-angiogenesis | Blocks tumor vascularization |
Table 2: Nanoparticle-based drug delivery systems in breast cancer
Combination Therapies
Combining drugs to tackle different pathways or overcome resistance. For example, a CDK4/6 inhibitor that blocks cell division can be combined with hormone therapy for hormone receptor-positive breast cancer [57]. Combining newer delivery systems with traditional chemotherapy or other treatments to enhance overall effectiveness. Implantable drug delivery devices are designed to release therapeutic agents over an extended period within the body, providing a continuous and localized dose of medication [58].
Role of WHO:
The WHO plays a central role in shaping the global breast cancer response by linking policy, data, technical guidance, and advocacy. Its Global Breast Cancer Initiative, supported by IARC’s data platforms and regional collaborations, offers a scalable framework to reduce mortality and narrow outcome disparities between high and low resource settings. The success of these efforts will depend on sustained national adoption, financing, and integration into broader cancer control and primary healthcare systems.
Global Breast Cancer Initiative (GBCI)
In 2021, WHO launched the Global Breast Cancer Initiative (GBCI), a flagship program that seeks to reduce global breast cancer mortality by 2.5% annually, to prevent 2.5 million deaths by 2040 [WHO, 2021]. The initiative rests on three interdependent pillars: (i) health promotion and early detection, aiming for ≥60% of invasive cancers to be diagnosed at stage I or II; (ii) timely diagnosis, with diagnostic confirmation ideally within 60 days of initial presentation; and (iii) comprehensive management, ensuring that at least 80% of patients complete full, guideline-recommended multimodal therapy [WHO, 2023].
Strategic Frameworks and National Integration
In 2023, the WHO introduced a roadmap for implementing the GBCI in diverse health system settings. This operational framework emphasizes embedding breast cancer control into primary healthcare and strengthening universal health coverage, particularly in resource-limited regions [WHO, 2023]. Regional adaptation has begun; for instance, in Punjab, India, the state government partnered with WHO to expand cancer care services across three districts, integrating screening, diagnostics, and treatment support at the community level.
Data and Technical Support through IARC
The International Agency for Research on Cancer (IARC), a specialized WHO body, provides the epidemiological backbone of the initiative. Through platforms such as the Global Cancer Observatory and the Global Initiative for Cancer Registry Development, IARC offers incidence, mortality, and survival data to guide policy and monitor progress. Tools like CanScreen5 and digital pathology atlases help standardize screening, diagnostics, and reporting, thereby supporting quality control and equitable access to services [IARC, 2022].
Addressing Inequalities
WHO emphasizes reducing global disparities in breast cancer outcomes. Five-year survival rates exceed 80–90% in high-income countries but fall below 40% in parts of sub-Saharan Africa [WHO, 2021]. Recent WHO reports highlight gaps in pathology infrastructure, workforce, and financing, with fewer than five African countries offering organized breast cancer screening. The GBCI roadmap prioritizes investment in workforce training, pathology expansion, and patient navigation programs to mitigate these inequities.
Advocacy and Classification
Beyond health systems strengthening, WHO engages in global advocacy campaigns such as Breast Cancer Awareness Month, emphasising patient-centred care under themes like “No one should face breast cancer alone” [WHO, 2024]. Additionally, through its “Blue Books” tumour classification series, WHO standardizes the histopathological categorization of breast tumours, ensuring diagnostic uniformity and facilitating international research collaboration
Future prospective
Breast cancer remains a major cause of mortality among women worldwide, posing significant treatment challenges due to its aggressive behaviour and limited targeted therapies. While conventional approaches such as surgery, radiotherapy, and chemotherapy have improved survival, issues like drug resistance and adverse side effects continue to limit their effectiveness. Breast cancer development is influenced by multiple risk factors, including genetic predisposition. Over the past two decades, treatment has shifted toward precision medicine based on molecular subtyping. Future strategies will emphasize individualized therapy, with treatment intensity adjusted according to tumour biology and predictive markers. However, the development of novel therapies for both early and advanced disease remains a challenge, with drug resistance being a key focus of ongoing research. Advances in single-cell technologies are improving understanding of tumour microenvironment interactions and may identify new biomarkers and therapeutic targets, such as CXCL13-positive T cells predicting response to anti-PD-L1 therapy in triple-negative breast cancer. Surgical approaches are also expected to become more biology-driven and personalised, although conducting randomized trials may be challenging due to safety concerns. Nanoparticle-based platforms help overcome biological barriers by enabling targeted tumour delivery, prolonged circulation, and controlled drug release. Future research should focus on optimising their design for personalised therapy, understanding nano–bio interactions, and exploring synergistic use with conventional treatments.
Conclusion
For control of breast cancer cases early diagnosis and clinical care are two important aspects. This could be possible by deciphering the molecular genetics and family history. Moreover, combinatorial approaches that integrate TME-targeted agents with conventional or molecularly tailored therapies may help overcome resistance and improve long-term outcomes. Therapeutic strategies which might target angiogenesis and successfully inhibit cell proliferation are to be used.Moving forward, a deeper understanding of the spatial and temporal dynamics of the TME, coupled with precision medicine approaches, will be essential for translating these insights into effective, patient-specific therapies. Ultimately, decoding and reprogramming the tumour microenvironment holds the potential not only to curb breast cancer progression but also to pave the way for durable therapeutic responses and improved survival. This realisation has led to the development of new therapeutic strategies for breast cancer treatment. led to the development of new therapeutic strategies for breast cancer treatment.
Recent advances in single-cell profiling, spatial transcriptomics, and in vivo modelling have illuminated the complexity of these interactions, offering a clearer view of the molecular and cellular players within the breast cancer ecosystem. Moving forward, a deeper understanding of the spatial and temporal dynamics of the TME, coupled with precision medicine approaches, will be essential for translating these insights into effective, patient-specific therapies. Ultimately, decoding and reprogramming the tumor microenvironment holds the potential not only to curb breast cancer progression but also to pave the way for durable therapeutic responses and improved survival.
References
- Kashyap D, Pal D, Sharma R, Garg VK, Goel N, Koundal D, et al. (2022). Global increase in breast cancer incidence: risk factors and preventive measures. Biomed Res Int. 2022:9605439.
View at Publisher | View at Google Scholar - Xiong X, Zheng LW, Ding Y, et al. (2025). Breast cancer: pathogenesis and treatments. Signal Transduct Target Ther. 10:49.
View at Publisher | View at Google Scholar - Menon G, Alkabban FM, Ferguson T. (2026). Breast cancer. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing.
View at Publisher | View at Google Scholar - Obeagu EI, Obeagu GU. (2024). Breast cancer: a review of risk factors and diagnosis. Medicine (Baltimore). 103(3):e36905.
View at Publisher | View at Google Scholar - Feng Y, Spezia M, Huang S, et al.(2018). Breast cancer development and progression: risk factors and molecular pathogenesis. Genes Dis. 5(2):77-106.
View at Publisher | View at Google Scholar - Zhang Y, Ji Y, Liu S, et al. (2025). Global burden of female breast cancer: estimates and projections to 2050. J Natl Cancer Cent. 5(3):287-296.
View at Publisher | View at Google Scholar - Freihat O, Sipos D, Kovacs A. (2025). Global burden and projections of breast cancer incidence and mortality. Front Public Health. 13:1622954.
View at Publisher | View at Google Scholar - Birnbaum JK, Duggan C, Anderson BO, Etzioni R. (2018). Early detection and treatment strategies for breast cancer in low-income and middle-income countries. Lancet Glob Health. 6(8): e885-e893.
View at Publisher | View at Google Scholar - Hoinoiu T, Piţ D, Oprean C, et al. (2025). Risk factors for breast cancer recurrence in postmenopausal women. Front Oncol. 15:1522713.
View at Publisher | View at Google Scholar - Katz TA. (2016). Mechanisms underlying the protective effect of pregnancy against breast cancer. Front Oncol. 6:228.
View at Publisher | View at Google Scholar - Al-Shami K, Awadi S, Khamees A, et al. (2023). Estrogens and breast cancer risk: a review. Heliyon. 9(9):e20224.
View at Publisher | View at Google Scholar - Clemente-Suarez VJ, Navarro-Jiménez E, Benítez-Agudelo JC, et al. (2025). Circadian disruption and cancer risk: a systematic review. J Natl Cancer Cent. 5(5):524-536.
View at Publisher | View at Google Scholar - Sun X, Qin L, Liang X, Wang D. (2026). Circadian rhythm in immunotherapy and tumor microenvironment. Acta Biochim Biophys Sin (Shanghai). 58(1):90-105.
View at Publisher | View at Google Scholar - Mohanty SS, Mohanty PK. (2019). Obesity as a breast cancer risk factor in postmenopausal women. Genes Dis.8(2):117-123.
View at Publisher | View at Google Scholar - Liu L, Hao X, Song Z, et al. (2021). Correlation between family history and breast cancer characteristics. Sci Rep.11:6360.
View at Publisher | View at Google Scholar - Petrucelli N, Daly MB, Pal T. (2026). BRCA1- and BRCA2-associated hereditary breast and ovarian cancer. GeneReviews® [Internet]. Seattle (WA): University of Washington.
View at Publisher | View at Google Scholar - Godet I, Gilkes DM. (2017). BRCA1 and BRCA2 mutations and treatment strategies. Integr Cancer Sci Ther. 4(1):1-10.
View at Publisher | View at Google Scholar - Sadeghi F, Asgari M, Matloubi M, et al. (2020). Molecular contribution of BRCA1/2 to genome instability. Biol Proced Online. 22:23.
View at Publisher | View at Google Scholar - Schrader KA, Masciari S, Boyd N, et al. (2011). Germline CDH1 mutations in breast cancer. J Med Genet. 48(1):64-68.
View at Publisher | View at Google Scholar - Khanabadi B, Najafgholizadeh Seyfi D, Rejali L, et al. (2023). STK11 mutation and cancer risk: a case study. Gastroenterol Hepatol Bed Bench. 16(3):341-346.
View at Publisher | View at Google Scholar - Fusco N, Sajjadi E, Venetis K, et al. (2020). PTEN alterations in cancer management. Genes (Basel). 11(7):719.
View at Publisher | View at Google Scholar - Orrantia-Borunda E, Anchondo-Nuñez P, Acuña-Aguilar LE, et al. (2022). Subtypes of breast cancer. In: Breast Cancer. Brisbane: Exon Publications.
View at Publisher | View at Google Scholar - Yersal O, Barutca S. (2014). Biological subtypes of breast cancer. World J Clin Oncol. 5(3):412-424.
View at Publisher | View at Google Scholar - Hu X, Chen W, Li F, et al. (2023). ER, PR, HER2 expression in breast cancer. Front Oncol. 13:1053125.
View at Publisher | View at Google Scholar - Onitilo AA, Engel JM, Greenlee RT, Mukesh BN. (2009). Breast cancer subtypes and clinical features. Clin Med Res. 7(1-2):4-13.
View at Publisher | View at Google Scholar - Pal SK, Childs BH, Pegram M. (2011). Triple-negative breast cancer: unmet needs. Breast Cancer Res Treat. 125(3):627-636.
View at Publisher | View at Google Scholar - Lei H, Fu J, Gu W, et al. (2026). Breast cancer: molecular pathogenesis and therapy. MedComm. 7(1):e70560.
View at Publisher | View at Google Scholar - Samad MA, Ahmad I, Khan MR, et al. (2025). Breast cancer: molecular pathogenesis and targeted therapy. MedComm. 6(10):e70404.
View at Publisher | View at Google Scholar - Thakur C, Qiu Y, Fu Y, et al. (2022). Epigenetics and environment in breast cancer. Front Oncol. 12:971288.
View at Publisher | View at Google Scholar - Pan L, Li J, Xu Q, et al. (2024). HER2/PI3K/AKT pathway in breast cancer. Medicine (Baltimore). 103(24):e38508.
View at Publisher | View at Google Scholar - Bouyahya A, Mechchate H, Oumeslakht L, et al. (2022). Epigenetic modifications in cancer. Biomolecules. 12(3):367.
View at Publisher | View at Google Scholar - Almazrouei KM, Mishra V, Pandya H, et al. (2025). Tumor microenvironment in cancer progression. Cureus. 17(9):e92707.
View at Publisher | View at Google Scholar - Wright K, Ly T, Kriet M, et al. (2023). Cancer-associated fibroblasts in tumor microenvironment. Cancers (Basel). 15(6):1899.
View at Publisher | View at Google Scholar - Huang R, Kang T, Chen S. (2024). Tumor-associated macrophages in immune evasion. J Cancer Res Clin Oncol. 150(5):238.
View at Publisher | View at Google Scholar - Zheng J, Hao H. (2024). Cancer-associated fibroblasts and drug resistance. Front Oncol. 13:1333839.
View at Publisher | View at Google Scholar - Lu J, Luo Y, Rao D, et al. (2024). Myeloid-derived suppressor cells in cancer. Exp Hematol Oncol. 13(1):39.
View at Publisher | View at Google Scholar - Emami Nejad A, Najafgholian S, Rostami A, et al. (2021). Hypoxia and cancer stem cells. Cancer Cell Int. 21:62.
View at Publisher | View at Google Scholar - Pitarch M, Alcantara R, Comerma L, et al. (2025). Imaging strategies for nipple discharge. Insights Imaging. 16(1):70.
View at Publisher | View at Google Scholar - Cerdas MG, Farhat J, Elshafie SI, et al. (2025). Evolution of breast cancer imaging. Cureus. 17(4):e82762.
View at Publisher | View at Google Scholar - Sebastian W, Forchette L, Donoughe K, et al. (2023). Hormone receptor-positive breast cancer. Cancers (Basel). 15(4):1303.
View at Publisher | View at Google Scholar - Wang J, Wu SG. (2023). Current therapeutic strategies in breast cancer. Breast Cancer (Dove Med Press). 15:721-730.
View at Publisher | View at Google Scholar - Palma M. (2025). Immunotherapy and cancer vaccines in breast cancer. Vaccines (Basel). 13(4):344.
View at Publisher | View at Google Scholar - Sriramulu S, Thoidingjam S, Speers C, et al. (2024). Immunotherapy for triple-negative breast cancer. Cancers (Basel). 16(19):3250.
View at Publisher | View at Google Scholar - Polgár C, Kahán Z, Ivanov O, et al. (2022). Radiotherapy of breast cancer. Pathol Oncol Res. 28:1610378.
View at Publisher | View at Google Scholar - Pandey S, Yadav P. (2025). MicroRNAs in cancer therapy. J Genet Eng Biotechnol. 23(4):100556.
View at Publisher | View at Google Scholar - Imani S, Wu RC, Fu J. (2018). MicroRNA-34 in breast cancer. J Cancer. 9(20):3765-3775.
View at Publisher | View at Google Scholar - He J, Zeng X, Wang C, et al. (2024). Antibody-drug conjugates in cancer therapy. MedComm. 5(8):e671.
View at Publisher | View at Google Scholar - Wang R, Hu B, Pan Z, et al. (2025). Antibody-drug conjugates: current and future. J Hematol Oncol. 18(1):51.
View at Publisher | View at Google Scholar - Yao Y, Zhou Y, Liu L, et al. (2020). Nanoparticle-based drug delivery in cancer therapy. Front Mol Biosci. 7:193.
View at Publisher | View at Google Scholar - Jenke R, Reßing N, Hansen FK, Aigner A, Büch T. (2021). Anticancer therapy with HDAC inhibitors: mechanism-based combination strategies and future perspectives. Cancers (Basel). 13(4):634.
View at Publisher | View at Google Scholar - Hurvitz SA, Dalenc F, Campone M, O'Regan RM, Tjan-Heijnen VC, et al. (2013). Everolimus combined with trastuzumab and paclitaxel in HER2-overexpressing advanced breast cancer: a phase 2 study. Breast Cancer Res Treat. 141(3):437-446.
View at Publisher | View at Google Scholar - Cord D, Rîmbu MC, Iordache MP, Albulescu R, Pop S, Tanase C, Popa ML. (2025). Phytochemicals as epigenetic modulators in chronic diseases: molecular mechanisms. Molecules. 30(21):4317.
View at Publisher | View at Google Scholar - Chan HJ, Petrossian K, Chen S. (2016). Structural and functional characterization of aromatase and estrogen receptor in endocrine-responsive and resistant breast cancer cells. J Steroid Biochem Mol Biol. 161:73-83.
View at Publisher | View at Google Scholar - Duro-Sánchez S, Alonso MR, Arribas J. (2023). Immunotherapies against HER2-positive breast cancer. Cancers (Basel). 15(4):1069.
View at Publisher | View at Google Scholar - Li J, Liu Y, Dai J, Yang L, Xiong F, Xia J, Jin J, Wu Y, Peng X. (2025). mRNA vaccines: current applications and future directions. MedComm. 6(11):e70434.
View at Publisher | View at Google Scholar - Lathia JD, Liu H. (2017). Overview of cancer stem cells and stemness. Target Oncol. 12(4):387-399.
View at Publisher | View at Google Scholar - Xue Y, Zhai J. (2024). Combining CDK4/6 inhibitors with other therapies and mechanisms of resistance. Int J Clin Exp Pathol. 17(7):189-207.
View at Publisher | View at Google Scholar - Pial MMH, Tomitaka A, Pala N, Roy U. (2022). Implantable devices for breast cancer treatment. J Nanotheranostics. 3(1):19-38.
View at Publisher | View at Google Scholar

 Clinic
Clinic
