Research Article | DOI: https://doi.org/10.31579/2835-9291/023
Detecting Individuals at High Risk of Liver Cirrhosis with Artificial Intelligence Deep Learning Algorithms
- Mehmet Kivrak *
Recep Tayyip Erdogan University Faculty of Medicine, Department of Biostatistics and Medical Informatics
*Corresponding Author: Mehmet Kivrak
Citation: Mehmet Kivrak, (2024), Detecting Individuals at High Risk of Liver Cirrhosis with Artificial Intelligence Deep Learning Algorithms, International Journal of Clinical Case Studies,3(4); DOI: 10.31579/2835-9291/023
Copyright: © 2024, Mehmet Kivrak. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Received: 04 July 2024 | Accepted: 22 July 2024 | Published: 31 July 2024
Keywords: liver cirrhosis; prediction level; deep learning
Abstract
The pathophysiology of liver cirrhosis reflects a complex interaction of many cellular and molecular mechanisms, and the development depends on a thorough understanding of this process of treatment and management strategies. The main objective of this machine learning project is to support healthcare professionals in making more precise treatment decisions and delivering personalized patient care by predicting a patient's cirrhosis stage using personal information such as medical history, age, gender, and other laboratory tests. Furthermore, with the assistance of an artificial intelligence-driven deep learning simulator, we can pinpoint individuals at an elevated risk of developing cirrhosis.
Methods: This project's Cirrhosis Prediction Dataset was obtained from Kaggle. The dataset includes patient ID, Day Count, Patient's Condition, Drug Type, Age, Gender, Ascites, Hepatomegaly, Spiders, Edema, Bilirubin, Cholesterol, Albumin, Copper, Alk_Phos (alkaline phosphatase), SGOT, Triglycerides, Platelets, Prothrombin, and Stage. We will perform predictive modeling using these data features an artificial intelligence-based deep learning algorithm was used to estimate the level of liver cirrhosis and develop a simulator that will enable early diagnosis.
Results: Within the deep learning prediction model, the markers that had the most significant impact on predicting liver cirrhosis level were assigned weights. According to Figure 7, the variable that made the most individual contribution was hepatomegaly (0.408), while the least contributing variable was n_days (0.001). Looking at the prediction chart, Clearly, the model produces higher accuracy predictions at the 3rd and 4th levels of liver cirrhosis. Model performance metrics were evaluated with Root Mean Squared Error (0.62), Absolute Error (0.51), Relative Error Lenient (18.3%) and Squared Error (0.46) values. In this case, it shows that overall, with this data the model performs reasonably well but could be improved or make better predictions.
Discussion: The success of our deep learning models carries the potential to diagnose patients earlier and treat them more effectively. Moreover, these models offer the potential to deliver personalized patient care and optimize resource utilization more efficiently. Our study offers valuable data for the early detection of the disease. Looking at the results of the model we developed, it generally shows that the model performs quite well with this data, but It's possible to enhance it for more accurate predictions.
Introduction
Liver cirrhosis, which is a worldwide public health issue, is a chronic and progressive liver condition marked by the substitution of healthy liver tissue with scar tissue (fibrosis), leading to compromised liver function. This condition can arise from various factors, including alcohol-related liver diseases, viral hepatitis infections, non-alcoholic fatty liver disease (NAFLD), metabolic syndrome, and autoimmune disorders [1-2]. This disease severely limits the liver's capacity to function normally and can lead to various complications. Liver cirrhosis is associated with serious consequences such as portal hypertension, intra-abdominal fluid accumulation (ascites or ascites), gastrointestinal bleeding, hepatic encephalopathy, and liver cancer. Additionally, liver cirrhosis can manifest itself with symptoms such as yellowing of the skin (as a result of impaired bile flow), muscle loss, fatigue, loss of appetite and weakness. [3-4]. Normal and cirrhotic liver organ images are given in figure 1.

Figure 1: Normal and Cirrhotic Liver Organ (4).
The pathophysiology of liver cirrhosis involves a intricate interplay of various cellular and molecular mechanisms, and comprehending this process is essential for the formulation of treatment and management approaches. Figure 2 illustrates the pathophysiology of cirrhosis. According to the figure, liver cirrhosis has been linked to several factors, chiefly chronic alcohol consumption, viral hepatitis infections (such as hepatitis B and hepatitis C), non-alcoholic fatty liver disease (NAFLD or NASH), autoimmune liver disorders, iron overload (hemochromatosis), and Wilson's disease [5-6]. Liver cirrhosis develops due to the abnormal reorganization of the liver resulting from prolonged damage [7]. This intricate pathophysiological
process involves a sequence of events and results in significant alterations in the structure and functionality of liver tissue. Initially, inflammation caused by chronic damage leads to inflammation of the liver tissue. This inflammation causes the death of hepatocytes (liver cells) and the migration of inflammatory cells (e.g., macrophages) to the area [8]. At the same time, regeneration of liver tissue begins to replace damaged hepatocytes. However, this process of regeneration also marks the beginning of a series of responses that form the basis of scar tissue accumulation. Instead of liver cells, cells such as fibroblasts begin to produce scar tissue [9]. This fibrotic response disrupts normal liver tissue and represents the onset of fibrosis.

Figure 2. The Pathophysiology of Cirrhosis
Fibrosis in liver tissue continues as fibroblasts produce excessive amounts of connective tissue components (e.g., collagen). The accumulation of this connective tissue disrupts the typical lobular structure of the liver, resulting in the onset of portal hypertension and influencing the flow of blood within the liver [10]. As a result, the vessels narrow and the pressure increases, which can lead to splenomegaly (spleen enlargement) and intra-abdominal fluid accumulation. As fibrosis and scar tissue accumulation in liver tissue progresses, liver functions are also seriously affected. This means that hepatocytes are unable to perform their functions and the liver's detoxification and metabolism abilities are reduced. At the same time, portal hypertension leads to the development of varicose veins, gastrointestinal bleeding, and an increased risk of liver cancer [11]. The diagnosis of this condition can be established through methods including clinical examination, laboratory tests (such as liver function tests), imaging studies (such as liver ultrasonography, CT scan, or MRI), liver biopsy, and elastography. Timely identification and the administration of suitable treatment can decelerate the advancement of the ailment and diminish the likelihood of complications.
The main objective of this machine learning project is to aid healthcare professionals in making more precise treatment decisions and delivering more individualized patient care by predicting a patient's cirrhosis stage based on personal information, including medical history, age, gender, and various laboratory tests. Additionally, with the help of an artificial intelligence-based deep learning simulator, we can identify individuals at a higher risk of developing cirrhosis.
2. Methods
2.1. Dataset
This project's Cirrhosis Prediction Dataset was obtained from Kaggle. The dataset contains various types of information about patients' medical history, laboratory test reports, and the stage of Liver Cirrhosis. The dataset contains 19 features, one target variable, and 418 observations. Cirrhosis is a condition where the liver is infected due to various liver diseases and disorders, including hepatitis and chronic alcoholism. The data in the dataset is gathered from research conducted on Primary Biliary Cirrhosis (PBC) during the period from 1974 to 1984 at the Mayo Clinic. You can find a complete description of the clinical research and all records in Chapter 0 of the book 'Counting Processes and Survival Analysis, Wiley, 1991' by Fleming and Harrington. Over the ten-year duration of the Mayo Clinic Trial, a total of 424 patients were included. The dataset includes patient ID, Day Count, Patient's Condition, Drug Type, Age, Gender, Ascites, Hepatomegaly, Spiders, Edema, Bilirubin, Cholesterol, Albumin, Copper, Alk_Phos (alkaline phosphatase), SGOT, Triglycerides, Platelets, Prothrombin, and Stage. We will perform predictive modeling using these data features [12].
Name | Explanation |
|
|
|
|
|
|
|
|
| |
ID | Unique Identifier. | ||||||||||
N_Days | number of days between registration and the earlier of death, transplantation, or study analysis time in July 1986. | ||||||||||
Status | Status of the patient C (censored), CL (censored due to liver tx), or D (death). | ||||||||||
Drug | Type of drug D-penicillamine or placebo. | ||||||||||
Age | age in [days]. | ||||||||||
Sex | M (male) or F (female). | ||||||||||
Ascites | Presence of Ascites N (No) or Y (Yes). | ||||||||||
Hepatomegaly | Presence of Hepatomegaly N (No) or Y (Yes). | ||||||||||
Spiders angiomas | Presence of Spiders N (No) or Y (Yes). | ||||||||||
Edema | presence of edema N (no edema and no diuretic therapy for edema), | ||||||||||
S (edema present without diuretics, or edema resolved by diuretics), or Y (edema despite diuretic therapy). | |||||||||||
Bilirubin | Serum Bilirubin in [mg/dl]. | ||||||||||
Cholesterol | Serum Cholesterol in [mg/dl]. | ||||||||||
Albumin | Albumin in [gm/dl]. | ||||||||||
Copper | Urine Copper in [ug/day]. | ||||||||||
Alk_Phos | Alkaline Phosphatase in [U/liter]. | ||||||||||
SGOT | SGOT in [U/ml]. | ||||||||||
Triglycerides | Triglicerides in [mg/dl]. | ||||||||||
Platelets | Platelets per cubic [ml/1000]. | ||||||||||
Prothrombin | prothrombin time in seconds [s]. |
|
|
|
|
|
|
| |||
Preprocessing
Table 1: Data Features

Figure 3: Data Preprocessing
2.2. Data Cleaning and
Random forest assignment method was used for missing values. 31 rows containing extreme/outlier values were detected in the data set. Based on the randomness test, these values were not deleted from the data set.
2.3. Deep Learning
Deep Learning possesses the ability to autonomously derive functional representations from raw data, presenting a pioneering approach to machine learning grounded in artificial neural networks [13]. Deep Learning learns hierarchical features by combining low-level features, effectively addressing complex and high-dimensional problems. Convolutional Neural Network (CNN) stands out as one of the most potent deep learning architectures [14]. The worth of the part of the layer l and its location within the k characteristic map (i, j),

can be evaluated as shown in equation 1.

=

(1)
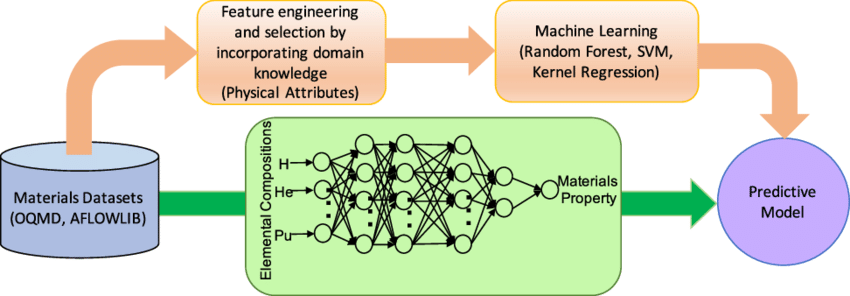
Figure 4: Comparison of Deep Learning Approac
The locations

and

are

th, lth layers within the k possession map are the weight vector and the bias. The activation value

for the convolution characteristic

can be stated, as shown in equation 2 (14).Liver cirrhosis, which is a worldwide public health issue, is a chronic and progressive liver condition marked by the substitution of healthy liver tissue with scar tissue (fibrosis), leading to compromised liver function. This condition can arise from various factors, including alcohol-related liver diseases, viral hepatitis infections, non-alcoholic fatty liver disease (NAFLD), metabolic syndrome, and autoimmune disorders [1-2]. This disease severely limits the liver's capacity to function normally and can lead to various complications. Liver cirrhosis is associated with serious consequences such as portal hypertension, intra-abdominal fluid accumulation (ascites or ascites), gastrointestinal bleeding, hepatic encephalopathy, and liver cancer. Additionally, liver cirrhosis can manifest itself with symptoms such as yellowing of the skin (as a result of impaired bile flow), muscle loss, fatigue, loss of appetite and weakness. [3-4]. Normal and cirrhotic liver organ images are given in figure 1.

Figure 1: Normal and Cirrhotic Liver Organ (4).
The pathophysiology of liver cirrhosis involves a intricate interplay of various cellular and molecular mechanisms, and comprehending this process is essential for the formulation of treatment and management approaches. Figure 2 illustrates the pathophysiology of cirrhosis. According to the figure, liver cirrhosis has been linked to several factors, chiefly chronic alcohol consumption, viral hepatitis infections (such as hepatitis B and hepatitis C), non-alcoholic fatty liver disease (NAFLD or NASH), autoimmune liver disorders, iron overload (hemochromatosis), and Wilson's disease [5-6]. Liver cirrhosis develops due to the abnormal reorganization of the liver resulting from prolonged damage [7]. This intricate pathophysiological
process involves a sequence of events and results in significant alterations in the structure and functionality of liver tissue. Initially, inflammation caused by chronic damage leads to inflammation of the liver tissue. This inflammation causes the death of hepatocytes (liver cells) and the migration of inflammatory cells (e.g., macrophages) to the area [8]. At the same time, regeneration of liver tissue begins to replace damaged hepatocytes. However, this process of regeneration also marks the beginning of a series of responses that form the basis of scar tissue accumulation. Instead of liver cells, cells such as fibroblasts begin to produce scar tissue [9]. This fibrotic response disrupts normal liver tissue and represents the onset of fibrosis.

Figure 2. The Pathophysiology of Cirrhosis
Fibrosis in liver tissue continues as fibroblasts produce excessive amounts of connective tissue components (e.g., collagen). The accumulation of this connective tissue disrupts the typical lobular structure of the liver, resulting in the onset of portal hypertension and influencing the flow of blood within the liver [10]. As a result, the vessels narrow and the pressure increases, which can lead to splenomegaly (spleen enlargement) and intra-abdominal fluid accumulation. As fibrosis and scar tissue accumulation in liver tissue progresses, liver functions are also seriously affected. This means that hepatocytes are unable to perform their functions and the liver's detoxification and metabolism abilities are reduced. At the same time, portal hypertension leads to the development of varicose veins, gastrointestinal bleeding, and an increased risk of liver cancer [11]. The diagnosis of this condition can be established through methods including clinical examination, laboratory tests (such as liver function tests), imaging studies (such as liver ultrasonography, CT scan, or MRI), liver biopsy, and elastography. Timely identification and the administration of suitable treatment can decelerate the advancement of the ailment and diminish the likelihood of complications.
The main objective of this machine learning project is to aid healthcare professionals in making more precise treatment decisions and delivering more individualized patient care by predicting a patient's cirrhosis stage based on personal information, including medical history, age, gender, and various laboratory tests. Additionally, with the help of an artificial intelligence-based deep learning simulator, we can identify individuals at a higher risk of developing cirrhosis.
2. Methods
2.1. Dataset
This project's Cirrhosis Prediction Dataset was obtained from Kaggle. The dataset contains various types of information about patients' medical history, laboratory test reports, and the stage of Liver Cirrhosis. The dataset contains 19 features, one target variable, and 418 observations. Cirrhosis is a condition where the liver is infected due to various liver diseases and disorders, including hepatitis and chronic alcoholism. The data in the dataset is gathered from research conducted on Primary Biliary Cirrhosis (PBC) during the period from 1974 to 1984 at the Mayo Clinic. You can find a complete description of the clinical research and all records in Chapter 0 of the book 'Counting Processes and Survival Analysis, Wiley, 1991' by Fleming and Harrington. Over the ten-year duration of the Mayo Clinic Trial, a total of 424 patients were included. The dataset includes patient ID, Day Count, Patient's Condition, Drug Type, Age, Gender, Ascites, Hepatomegaly, Spiders, Edema, Bilirubin, Cholesterol, Albumin, Copper, Alk_Phos (alkaline phosphatase), SGOT, Triglycerides, Platelets, Prothrombin, and Stage. We will perform predictive modeling using these data features [12].
Name | Explanation |
|
|
|
|
|
|
|
|
| |
ID | Unique Identifier. | ||||||||||
N_Days | number of days between registration and the earlier of death, transplantation, or study analysis time in July 1986. | ||||||||||
Status | Status of the patient C (censored), CL (censored due to liver tx), or D (death). | ||||||||||
Drug | Type of drug D-penicillamine or placebo. | ||||||||||
Age | age in [days]. | ||||||||||
Sex | M (male) or F (female). | ||||||||||
Ascites | Presence of Ascites N (No) or Y (Yes). | ||||||||||
Hepatomegaly | Presence of Hepatomegaly N (No) or Y (Yes). | ||||||||||
Spiders angiomas | Presence of Spiders N (No) or Y (Yes). | ||||||||||
Edema | presence of edema N (no edema and no diuretic therapy for edema), | ||||||||||
S (edema present without diuretics, or edema resolved by diuretics), or Y (edema despite diuretic therapy). | |||||||||||
Bilirubin | Serum Bilirubin in [mg/dl]. | ||||||||||
Cholesterol | Serum Cholesterol in [mg/dl]. | ||||||||||
Albumin | Albumin in [gm/dl]. | ||||||||||
Copper | Urine Copper in [ug/day]. | ||||||||||
Alk_Phos | Alkaline Phosphatase in [U/liter]. | ||||||||||
SGOT | SGOT in [U/ml]. | ||||||||||
Triglycerides | Triglicerides in [mg/dl]. | ||||||||||
Platelets | Platelets per cubic [ml/1000]. | ||||||||||
Prothrombin | prothrombin time in seconds [s]. |
|
|
|
|
|
|
| |||
Preprocessing
Table 1: Data Features

Figure 3: Data Preprocessing
2.2. Data Cleaning and
Random forest assignment method was used for missing values. 31 rows containing extreme/outlier values were detected in the data set. Based on the randomness test, these values were not deleted from the data set.
2.3. Deep Learning
Deep Learning possesses the ability to autonomously derive functional representations from raw data, presenting a pioneering approach to machine learning grounded in artificial neural networks [13]. Deep Learning learns hierarchical features by combining low-level features, effectively addressing complex and high-dimensional problems. Convolutional Neural Network (CNN) stands out as one of the most potent deep learning architectures [14]. The worth of the part of the layer l and its location within the k characteristic map (i, j),

can be evaluated as shown in equation 1.

=

(1)
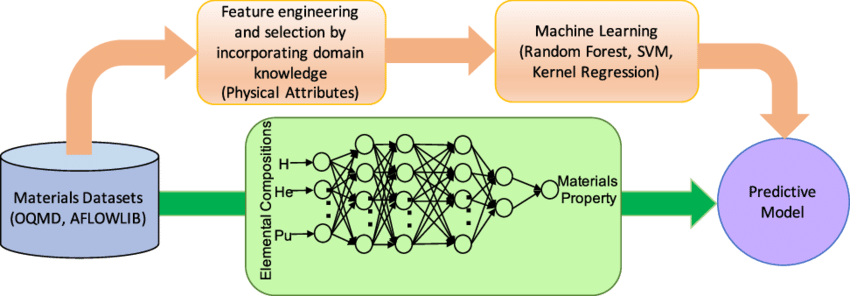
Figure 4: Comparison of Deep Learning Approac
The locations

and

are

th, lth layers within the k possession map are the weight vector and the bias. The activation value

for the convolution characteristic

can be stated, as shown in equation 2 (14).
Results
In the deep learning algorithm, scoring date information consists of epoch iteration, training sample, RMSE, deviation, MAE and r2 values. After each epoch, the model compares its predictions with the actual labels and tries to minimize the errors resulting from this comparison. Increasing the number of epochs allows the model to do more learning and better adapt to the training data. A lower RMSE and deviance values indicate that the model's predictions are closer to the actual values and perform better. So, 0.62 and 0.38 can be considered a low RMSE and deviance values. Detailed scoring history is shown in figure 5.

Figure 5: Scoring History in Deep Learning
In the deep learning approach designed as a regression model, model hyperparameters were set as weights/biases: 4.7, 2510 training samples and mini-batch size: 1 in gaussian distribution. The detailed deep learning technique is illustrated in Figure 6.

Figure 6: Detailed Deep Learning Approach
In the deep learning prediction model, the markers that contributed the most to the liver cirrhosis prediction level were weighted. According to Figure 7, the variable that made the most individual contribution was hepatomegaly (0.408), while the variable that had the smallest impact was n_days (0.001).

Figure 7: Attribute Weight
The prediction chart shows how the model predicts the data points. Predictions are usually shown as a line, curve, or dot. Through the analysis of the correlation between observed values and predictions, it enables us to discern which data points the model accurately forecasted and which it inaccurately forecasted. Analyzing the graph in Figure 8, it becomes evident that the model yields higher accuracy predictions for the 3rd and 4th stages of liver cirrhosis.

Figure 8: Prediction Chart
According to Table 2, model performance metrics were evaluated with Root Mean Squared Error (0.62), Absolute Error (0.51), Relative Error Lenient (18.3%) and Squared Error (0.46) values. In this case, it shows that overall, with this data the model performs reasonably well but could be improved or make better predictions.
Model | |||
Deep Learning | Performans Metric | Value | 95 % CI |
Root Mean Squared Error | 0.62 | (0.59-0.69) | |
Absolute Error | 0.51 | (0.43-0.59) | |
Relative Error Lenient (%) | 18.3 | (16.4-20.1) | |
| Squared Error | 0.46 | (0.35-0.57) |
Table 2: Model Performance Metrics
In the last step, liver cirrhosis level is estimated based on the parameters determined by the simulation model, and it helps in the clinical decision process by diagnosing individuals at an early stage. In the representation given in Figure 9, a liver cirrhosis level was estimated with user-entered parameter values (3.393). This value means that the patient presenting with the clinical marker values used in the simulator is in a level 3 disease risk group and the treatment protocol should be shaped accordingly.

Figure 9: Deep Learning Simulator
Discussion
In recent years, deep learning methods have garnered significant interest in the healthcare industry, and they have assumed a crucial role in medical diagnosis and predictive studies. Esteva et al. (2019) pioneered the development of deep learning models utilizing a substantial patient dataset, training these models for the prediction of liver cirrhosis levels. The data set included patients' medical histories, laboratory results, and imaging studies. By processing these diverse data, deep learning methods have achieved high success rates for predicting the level of liver cirrhosis [15].
Litjens et al. (2017) conducted a study on predicting liver cirrhosis levels to assess the impact of deep learning algorithms in clinical applications [16].
Johnson et al. (2019) achieved success in predicting the stages of liver cirrhosis by efficiently utilizing deep learning models and various data types. Their findings demonstrated that deep learning methods can serve as a potent tool for the prediction and diagnosis of complex diseases [17].
The success of our deep learning models carries the potential to diagnose patients earlier and treat them more effectively. Additionally, these models offer the opportunity to provide individualized patient care and use resources more effectively. Estimating the level of a complex disease such as liver cirrhosis can help make better and more informed clinical decisions. However, using the deep learning approach in medical applications also poses some difficulties. Matters like the requirement for extensive datasets, the time and resources needed for model training, the transparency and reliability of algorithms are crucial considerations that require attention. Furthermore, it's important to consider the confidentiality of patients' data and address ethical concerns in the process.
Our study furnishes valuable data for the early diagnosis of the disease. Evaluating the outcomes of the model we have developed; it generally demonstrates satisfactory performance with this dataset. However, there is room for improvement, and more accurate predictions can potentially be achieved.
In conclusion, deep learning methods have great potential in medical studies such as liver cirrhosis level prediction and similar medical studies. However, further research, development and standardization studies are needed to successfully integrate these technologies into clinical applications.
References
- Tsochatzis, E. A., Bosch, J., & Burroughs, A. K. (2014). Liver cirrhosis. The Lancet, 383(9930), 1749-1761.
View at Publisher | View at Google Scholar - Younossi, Z., Tacke, F., Arrese, M., Sharma, B. C., Mostafa, I., Bugianesi, E., ... & Vos, M. B. (2019). Global perspectives on nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology, 69(6), 2672-2682.
View at Publisher | View at Google Scholar - Manns, M., Samuel, D., Gane, E. J., Mutimer, D., McCaughan, G., Buti, M., ... & Forns, X. (2016). Ledipasvir and sofosbuvir plus ribavirin in patients with genotype 1 or 4 hepatitis C virus infection and advanced liver disease: a multicentre, open-label, randomised, phase 2 trial. The Lancet Infectious Diseases, 16(6), 685-697.
View at Publisher | View at Google Scholar - https://surgery.ucsf.edu/conditions--procedures/cirrhosis.aspx
View at Publisher | View at Google Scholar - Younossi, Z. M., Loomba, R., Anstee, Q. M., Rinella, M. E., Bugianesi, E., Marchesini, G., ... & Lindor, K. (2018). Diagnostic modalities for nonalcoholic fatty liver disease, nonalcoholic steatohepatitis, and associated fibrosis. Hepatology, 68(1), 349-360.
View at Publisher | View at Google Scholar - Asrani, S. K., Devarbhavi, H., Eaton, J., & Kamath, P. S. (2019). Burden of liver diseases in the world. Journal of hepatology, 70(1), 151-171.
View at Publisher | View at Google Scholar - Schuppan, D., & Afdhal, N. H. (2008). Liver cirrhosis. The Lancet, 371(9615), 838-851.
View at Publisher | View at Google Scholar - Seki, E., & Schwabe, R. F. (2015). Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology, 61(3), 1066-1079.
View at Publisher | View at Google Scholar - Iredale, J. P., Thompson, A., & Henderson, N. C. (2013). Extracellular matrix degradation in liver fibrosis: biochemistry and regulation. Biochimica et Biophysica Acta (BBA)-Molecular Basis of Disease, 1832(7), 876-883.
View at Publisher | View at Google Scholar - Friedman, S. L. (2008). Hepatic fibrosis—overview. Toxicology, 254(3), 120-129.
View at Publisher | View at Google Scholar - Barr, R. G., Ferraioli, G., Palmeri, M. L., Goodman, Z. D., Garcia-Tsao, G., Rubin, J., ... & Levine, D. (2015). Elastography assessment of liver fibrosis: society of radiologists in ultrasound consensus conference statement. Radiology, 276(3), 845-861.
View at Publisher | View at Google Scholar - https://www.kaggle.com/datasets/fedesoriano/cirrhosis-prediction-dataset
View at Publisher | View at Google Scholar - Li, Y., Nie, X., & Huang, R. (2018). Web spam classification method based on deep belief networks. Expert Systems with Applications, 96, 261-270.
View at Publisher | View at Google Scholar - Gu, J., Wang, Z., Kuen, J., Ma, L., Shahroudy, A., Shuai, B., ... & Chen, T. (2018). Recent advances in convolutional neural networks. Pattern Recognition, 77, 354-377.
View at Publisher | View at Google Scholar - Esteva, A., Robicquet, A., Ramsundar, B., Kuleshov, V., DePristo, M., Chou, K., ... & Dean, J. (2019). A guide to deep learning in healthcare. Nature medicine, 25(1), 24-29.
View at Publisher | View at Google Scholar - Litjens, G., Kooi, T., Bejnordi, B. E., Setio, A. A. A., Ciompi, F., Ghafoorian, M., ... & Sánchez, C. I. (2017). A survey on deep learning in medical image analysis. Medical image analysis, 42, 60-88.
View at Publisher | View at Google Scholar - Johnson, J. M., & Khoshgoftaar, T. M. (2019). Survey on deep learning with class imbalance. Journal of Big Data, 6(1), 1-54.
View at Publisher | View at Google Scholar

 Clinic
Clinic
